Figures & data
Figure 1. Regional hyperthermia as targeted therapy. Schematic illustration of regional hyperthermia (39–43 °C) targeting the heating field to the area of an intra-abdominal, deep-seated malignant tumour. The external heating device is represented by an open applicator surrounding the body axis of the patient. The increased understanding to control energy deposition in vivo has led to the design of commercially available multi-antenna applicators and the implementation of systems for monitoring temperature distribution within the heated tumour and the tumour-associated stroma.
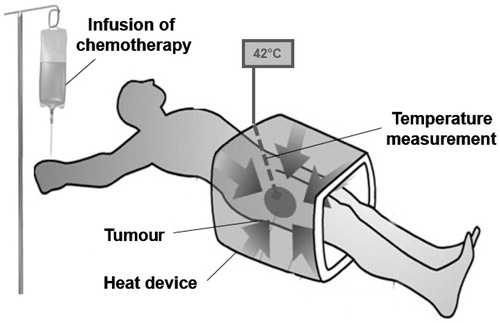
Figure 2. The hallmarks of hyperthermia. This illustration encompasses six hallmark capabilities induced by heat. The past two decades have witnessed fundamental discoveries toward understanding the mechanistic insights of each hallmark and the linkage to each other.
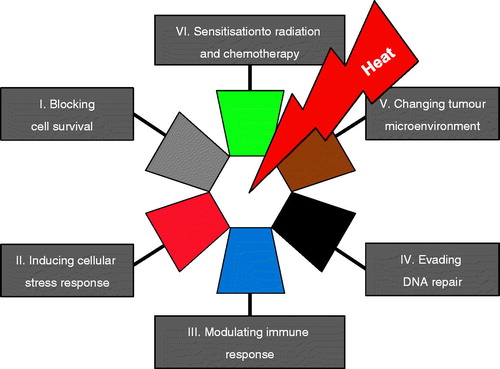
Figure 3. Trabectedin isolated from the Caribbean marine tunicate - an anticancer agent. The unique mechanisms of action include the initial formation of DNA–trabectedin–XPG ternary complexes. The endonuclease ERRC5 (xeroderma pigmentosum group G; XPG human; Rad13 yeast) is a key-protein of the DNA nucleotide excision repair (NER) pathway. Photos courtesy of PharmaMar.
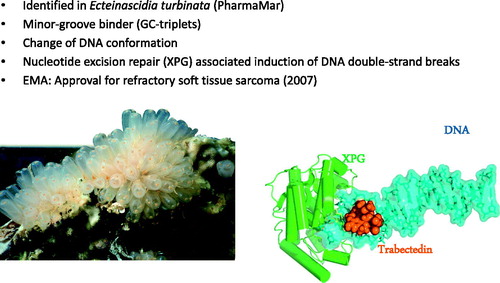
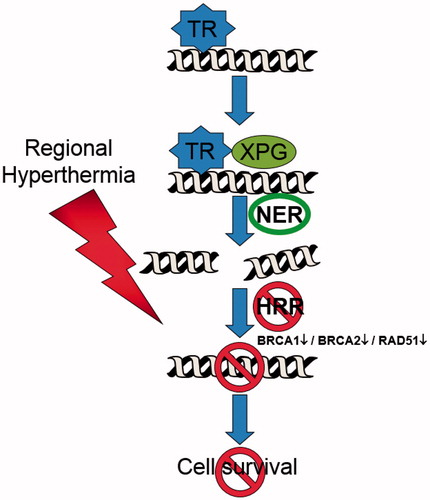
Figure 4. Regional hyperthermia targeting the homologous recombination repair (HRR) pathway. In combination with trabectedin, the formation of the DNA–trabectedin–XPG complex triggers a cascade of events, which ultimately lead to the generation of DSBs. The HRR pathway is inhibited over a sustained period of time after the application of regional hyperthermia due to an immediate degradation of BRCA2 and – later on – of BRCA1 resulting in an increase in unresolved DSBs and cell death.