Figures & data
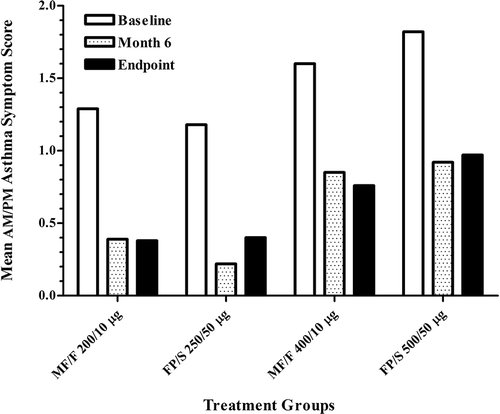
Figure 1. —Patient disposition. ICS, inhaled corticosteroid; FP/S, fluticasone propionate/salmeterol combination; MF/F, mometasone furoate/formoterol. *Patient population used for safety analyses. †Patient population used for efficacy analyses, identified before database lock.
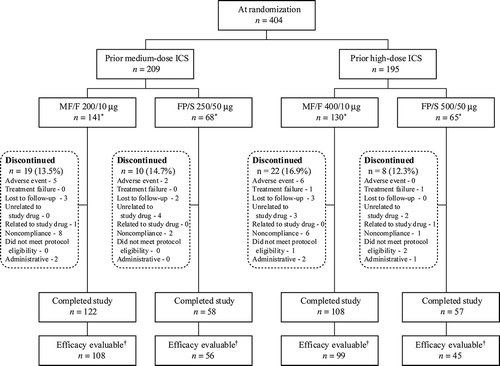
Table 1. —Patient demographics and baseline characteristics
Table 2. —Most common treatment-emergent adverse events reported by ≥5.0% of patients in any treatment group
Table 3. —Most common treatment-related adverse events reported by ≥2.0% of patients in any treatment group
Figure 2. —Mean plasma cortisol 24-hour AUC at baseline, week 26, and week 52. Patients who had both 0- and 24-hour measurements were included in the analysis. AUC, area under the curve; FP/S, fluticasone propionate/salmeterol; MF/F, mometasone furoate/formoterol.
*p≤ .043 versus baseline within treatment groups.
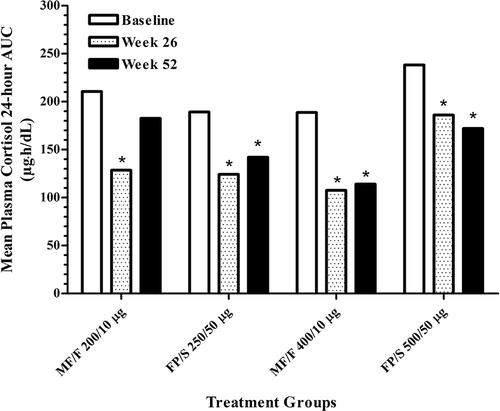
Figure 3. —Improvement in mean 24-hour total asthma symptom scores. Total asthma symptom scores are the sum of wheezing, coughing, and difficulty breathing scores, based on a scale of 0 (none) to 3 (severe). FP/S, fluticasone propionate/salmeterol; MF/F, mometasone furoate/formoterol.