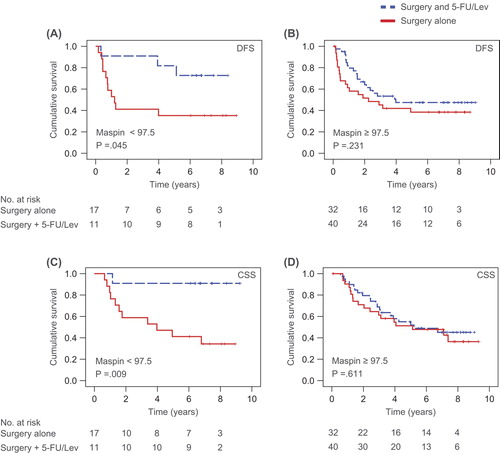
Figures & data
Table I. Patient characteristics and maspin expression for patients included in the analyses.
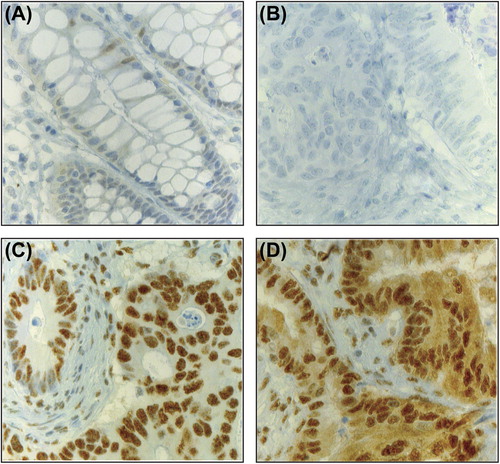
Table II. Nuclear maspin expression score according to patient and tumor characteristics.
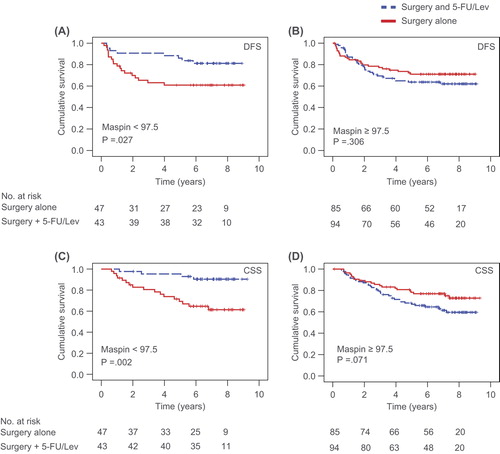
Table III. Univariate Cox regression analysis for cancer-specific survival.