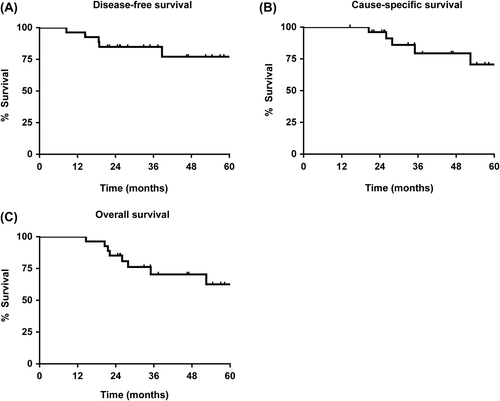
Figures & data
Table I. Baseline patient and disease characteristics.
Table II. Maximum physician recorded acute toxicity (CTCAE version 3) during treatment (number of patients, percentage of total in parentheses).
Table III. Maximum physician recorded late toxicity (based on CTCAE version 3.0) from 6 months after treatment (number of patients, percentage of total in parentheses).
Table IV. Patient-reported late toxicity (based on CTCAE version 3.0) from 6 months after treatment (number of patients and percentage of total).
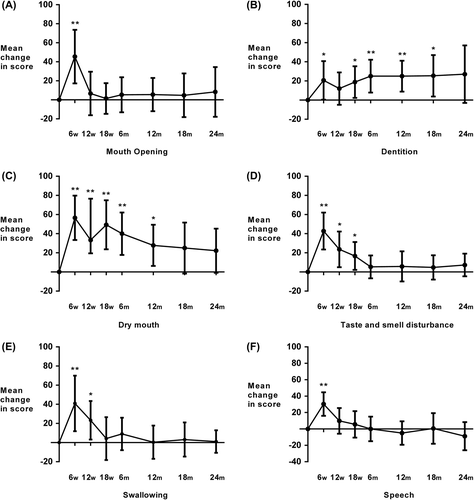
Figure 1. Patient-reported (EORTC QLQ–H&N35 questionnaire) change in mean symptoms scores from baseline at 6, 12 and 18 weeks from start of treatment and 6, 12, 18 and 24 months from completion of treamtment for A. mouth opening, B. dentition, C. dry mouth, D. taste and smell, D. swallowing, E.speech. Statistical comparisons are versus baseline scores, *p< 0.01, **p < 0.001; error bars represent 99% confidence intervals. w, weeks; m, months.