Figures & data
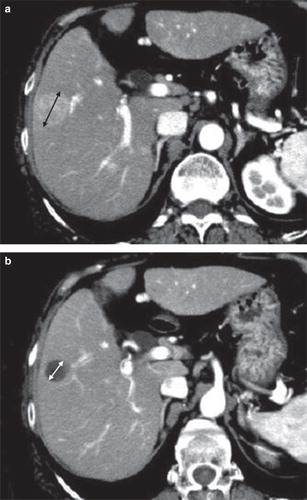
Figure 1. A: RECIST ver. 1.1: Response was defined according to a unidimensional measurement of the entire lesion, including the necrotic part. B: mRECIST: Response was defined according to a unidimensional measurement of the viable part, excluding the necrotic part.
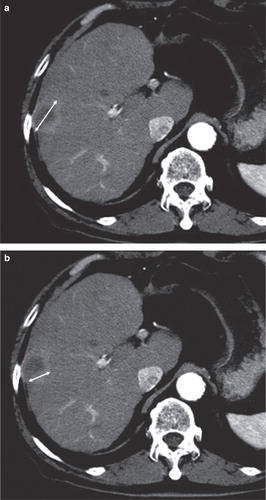
Table I. Patients and characteristics.
Table II. Inter-criteria reproducibility between RECIST version 1.1 and mRECIST criteria. Number of lesions (%).
Table III. Inter-criteria reproducibility between RECIST version 1.1 and mRECIST criteria: distribution chart.
Table IV. Inter-observer reproducibility.
Table V. Intra-observer reproducibility.
Figure 2. A: CT before TACE: Both criteria (RECIST version 1.1 and mRECIST) measured the longest diameter of the tumor. B: CT after TACE: The tumor had become entirely necrotic. The tumor response was evaluated as CR using mRECIST criteria (i.e. no measurement) and as non-CR using RECIST version 1.1 criteria (i.e. the measurement of the longest diameter of the entire tumor).