Figures & data
Table I. Guidance on laboratory assays to measure rivaroxaban in plasma.
Figure 1. Prothrombin time (PT) measured A: in seconds (Citation13); and B: as INR (Lindhoff-Last, unpublished data) in patients undergoing hip or knee replacement surgery taking rivaroxaban 10 mg once daily. Results are median values (n = 47). INR = international normalized ratio.
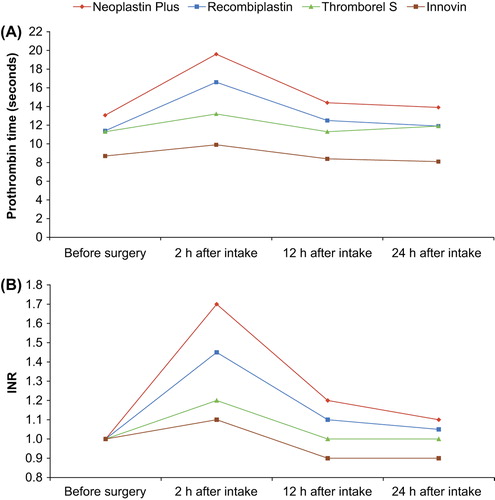
Figure 2. Transitioning from rivaroxaban to a vitamin K antagonist (VKA). *Approved rivaroxaban dose for treatment of deep vein thrombosis (DVT), and prevention of recurrent DVT and pulmonary embolism following an acute DVT, and for the prevention of stroke and systemic embolism in patients with non-valvular atrial fibrillation. INR = international normalized ratio.
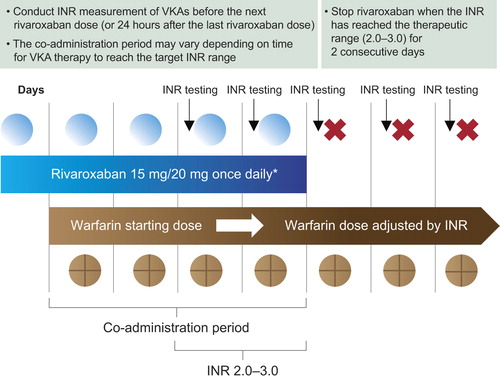
Table II. Plasma concentrations of rivaroxaban at steady state in different patient populations.