Figures & data
Figure 2. Light microscopic appearances of inherited skin fragility at either the dermal-epidermal junction (A) or within the lower epidermis (B). Asterisk indicates the blister formation (Richardson's stain; scale bar = 50 μm).
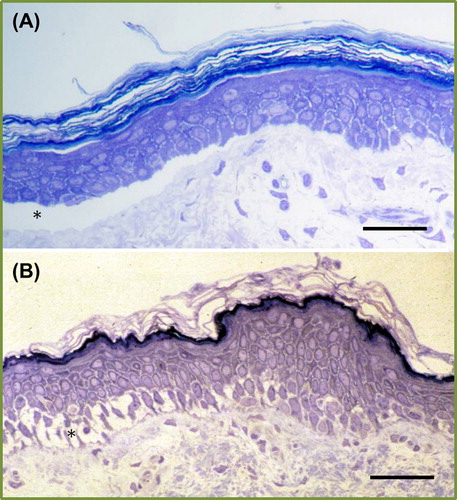
Figure 3. Stylized diagram depicting the important components in the skin that provide the keratinocyte, cell-cell, and cell-extracellular matrix integrity along with the protein components that are mutated in inherited skin fragility diseases.
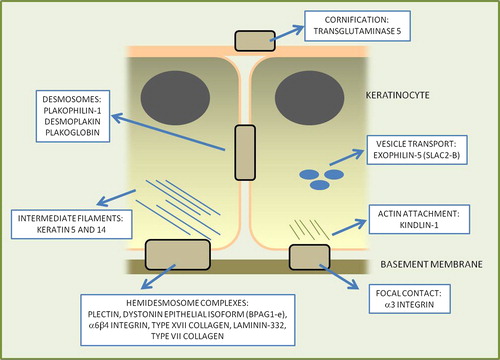
Figure 4. Clinical illustrations of different genetic disorders showing varying degrees of skin fragility. In each case the mutated skin protein is shown.
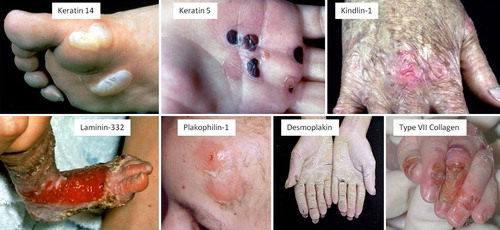
Table I. Summary of the gene and protein targets and disease phenotypes in the inherited skin fragility disorders.
Figure 5. Clinical illustration of the therapeutic challenge presented by a patient with recessive dystrophic EB and very extensive skin fragility. The various endeavours to develop new therapies that are currently at preclinical or clinical trial stages are listed.
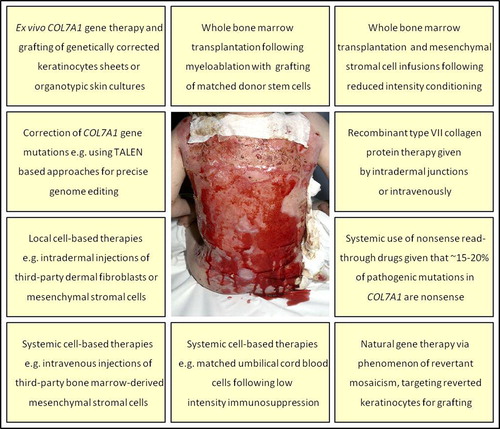
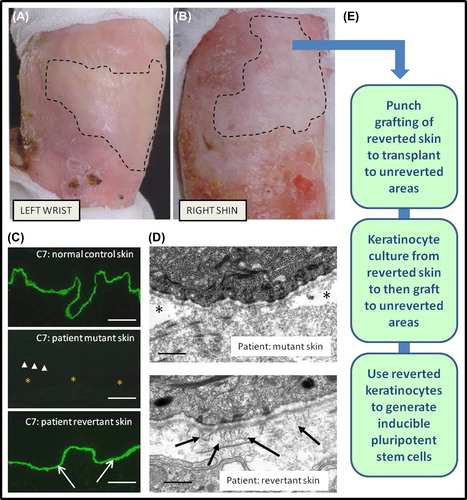
Figure 6. Clinicopathological features of revertant mosaicism in a patient with recessive dystrophic EB and potential clinical translation for therapy. There are demarcated areas in which trauma-induced blistering does not occur on the left wrist (A) and right shin (B). Immunolabelling of the dermal-epidermal junction (C) with an antibody to type VII collagen (C7) shows bright linear staining in normal skin but a complete absence in patient skin (arrow-heads indicate dermal-epidermal junction, and asterisk denotes subepidermal blistering). In contrast, in the revertant areas there is bright linear labelling for type VII collagen (arrows) which resembles normal skin (scale bar = 50 μm). Ultrastructurally (D), the patient's blister-prone skin shows focal blistering beneath the lamina densa (asterisk), whereas in the area of revertant mosaicism there is no blistering and also evidence of new anchoring fibrils (arrows) that are completely lacking in the blistered skin (scale bar = 0.2 μm). The phenomenon of revertant mosaicism presents several opportunities for translational research and therapy (E), including culturing the reverted keratinocytes, skin grafting, and creation of inducible pluripotent stem cells.