Figures & data
TABLE 1. Population demographics and baseline characteristics (efficacy population).
TABLE 2. Change from baseline in composite blepharitis severity at all study visits (ITT population).
TABLE 3. Proportion of all study eyes with a ≥1-grade reduction (improvement) from baseline to Day 15 in blepharitis signs (ITT population).
FIGURE 1. Mean individual blepharitis sign severity at each study visit. Entirety of stacked bar depicts composite severity. Severity of each sign rated on a 5-point scale of 0 (none) to 4 (severe). LE/T, loteprednol etabonate 0.5%/tobramycin 0.3%; DM/T, dexamethasone 0.1%/tobramycin 0.3%
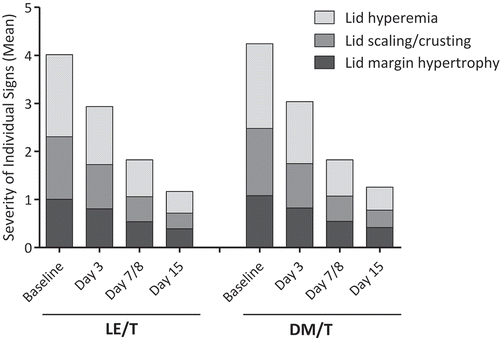
FIGURE 2. Percentages of patients (eyes) whose blepharitis signs were resolved (i.e., grade 0) at Day 15. LE/T, loteprednol etabonate 0.5%/tobramycin 0.3%; DM/T, dexamethasone 0.1%/tobramycin 0.3%
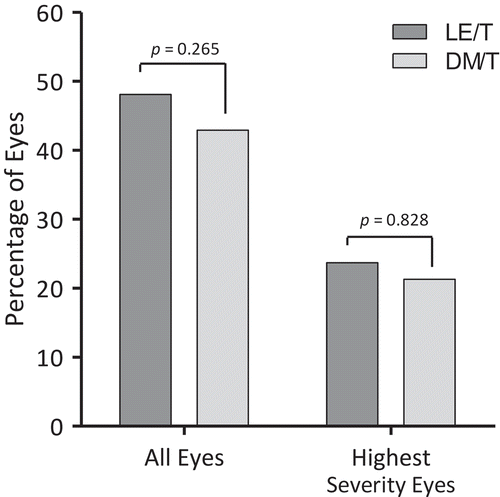
TABLE 4. Treatment emergent ocular adverse events (study eye).
FIGURE 3. IOP changes from baseline in the (A) US population and (B) Chinese population. LE/T, loteprednol etabonate 0.5%/tobramycin 0.3%; DM/T, dexamethasone 0.1%/tobramycin 0.3%.
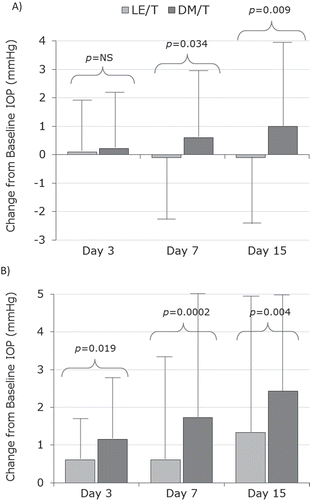
TABLE 5. Proportion of patients with a ≥5 mmHg and ≥10 mmHg increase in IOP.
