Figures & data
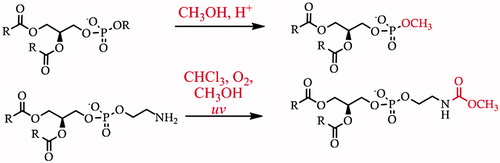
Figure 1. Relative polarities of biological lipid species, (A) hexadecanol/cetyl alcohol, (B) trioleoyl glycerol, (C) cholesterol, (D) dioleoyl phosphatidylcholine, (E) phosphatidylinositol-3,4,5-tris-phosphate, (F) a generic lipopolysaccharide. The blue and red dashed lines show an approximate delineation between the head group region, the connecting region, and the hydrophobic fraction. The black dotted lines show an approximate delineation between lipophilic and intermediate (left) and intermediate and hydrophilic (right) lipid species.
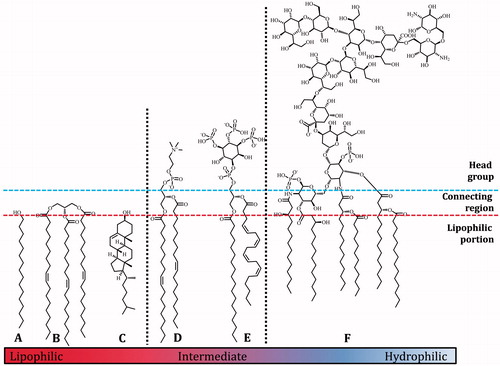
Scheme 1. Two types of chemical modification that may be observed in handling lipids (Garrett et al., Citation2011). Upper, acid-mediated electrophilic substitution of the phosphate di-ester that produces non-endogenous lipids such as phosphatidylmethanol. Lower, the degradation of chloroform on storage can produce phosgene that in turn may lead to carbamoylation of PE.