Figures & data
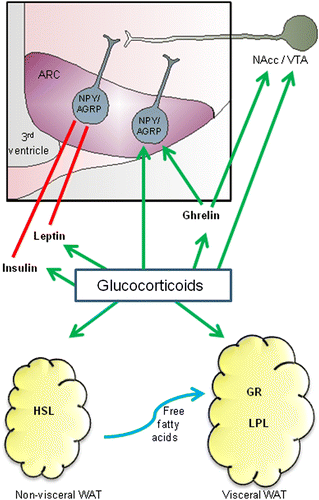
Figure 1. An overview of the concepts discussed for GC regulation of appetite and adiposity. GCs stimulate food intake via direct action on orexigenic NPY/AGRP neurons in the ARC, as well by stimulating nucleus accumbens (NAcc)/VTA pathways that reinforce the rewarding nature of food. GC may also stimulate food intake by enhancing the appetite- and reward-stimulatory effects of ghrelin. In addition, GC can prevent the actions of satiety hormones leptin and insulin; reducing the sensitivity of the brain to these hormones and contributing to leptin and insulin resistance. GC also contributes to enhancing visceral fat WAT. They activate HSL, enhancing lipolysis and LPL, promoting fat storage. Because there is a greater density of GRs in visceral fat, as well as greater LPL activity, fat storage may be preferentially promoted in this tissue. See text for details.