Figures & data
Table 1. Composition of the lipid phase of the different kinds of vesicular systems examined.
Table 2. Mean vesicle size (n = 6), polydispersity index (PDI) (n = 6) and entrapment efficiency (EE%) (n = 5) of the different CXB-loaded (0.5% w/v) vesicular dispersions.
Figure 1. Transmission electron micrographs of liposomes (batch A), transfersomes (batch D) and ethosomes (batch F; see and for batch formulation composition).
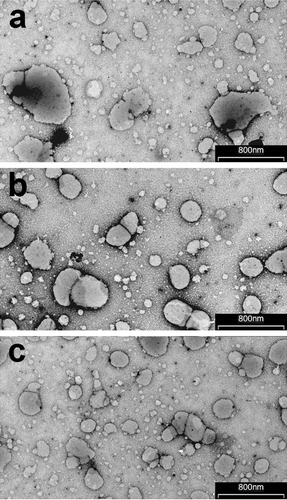
Table 3. Mean vesicle size, polydispersity index (PDI) and entrapment efficiency (EE%) of the selected CXB-loaded (0.5% w/v) vesicular systems after 30 days storage at 4°C and room temperature (25°C).
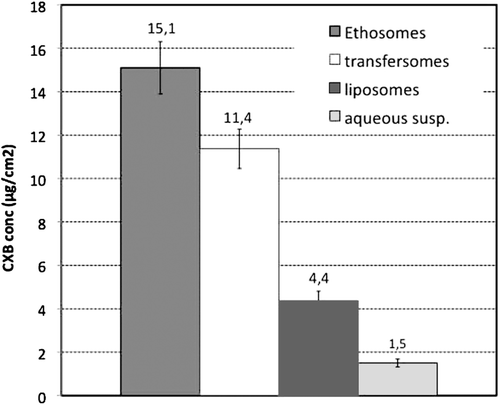
Figure 2. Amount of Celecoxib (CXB) penetrated into the excised human skin after 4 h from application of the three kinds of selected vesicular formulations (liposomes, batch A; transfersomes, batch D; ethosomes, batch F) and of an aqueous drug suspension as reference (see and for batch formulation composition; each result is the mean of five separate experiments).