Figures & data
Table 1. Anatomy and physiology of vagina.
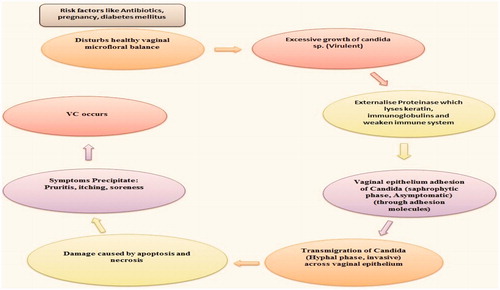
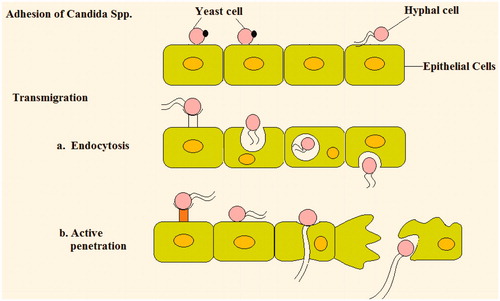
Table 2. Ligand specific adhesion and invasions involved in adhesion and transmigration across vaginal epithelium.
Table 3. Worldwide distribution of various Candida sp. responsible for vaginal candidiasis.
Table 4. Factor affecting vaginal drug absorption.
Table 6. Drug associated limitations for anti-fungal therapy.
Table 7. Vaginal bio-adhesive tablets loaded with anti-fungal agent.
Table 8. Anti-fungal agents formulated as vaginal liposomes.
Table 9. Micro-emulsion based vaginal gel loaded with anti-fungal agents.
Table 10. Bio-adhesive gel loaded with different anti-fungal agents.
Table 11. Different anti-fungal vaginal micro particle formulations.
Table 12. Recently developed vaccines against VC.