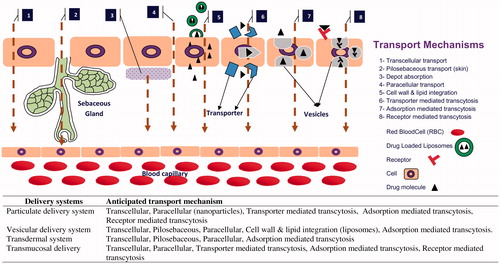
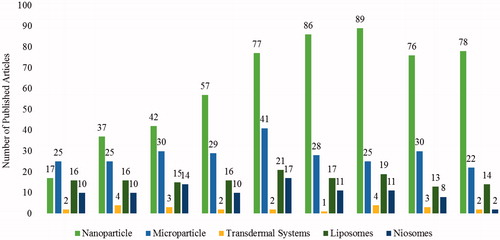
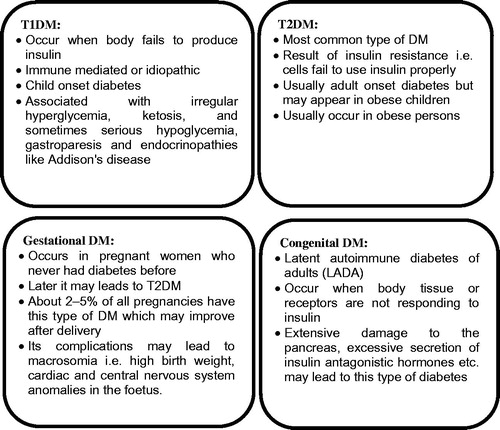
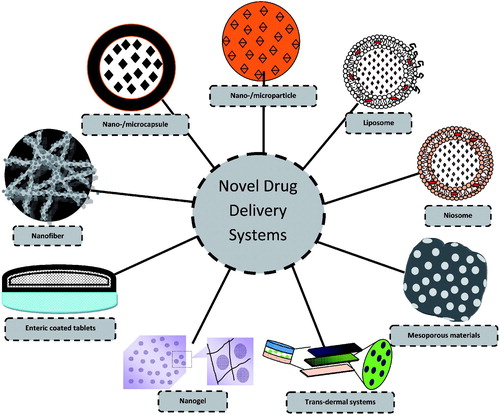
Figures & data
Table 1. Some antidiabetic drugs in conventional formulations.
Table 2. Novel drug delivery systems for antidiabetics – advantages and limitations.
Table 3. Recent reports on various microparticulate systems for the treatment of diabetes (Year 2005 onward).
Table 4. Recent reports on different nanoparticulate approaches used for antidiabetics (Year 2005 onward).
Table 5. Vesicular systems developed for antidiabetic drugs.
Table 6. Miscellaneous noteworthy novel carriers for antidiabetics.