Figures & data
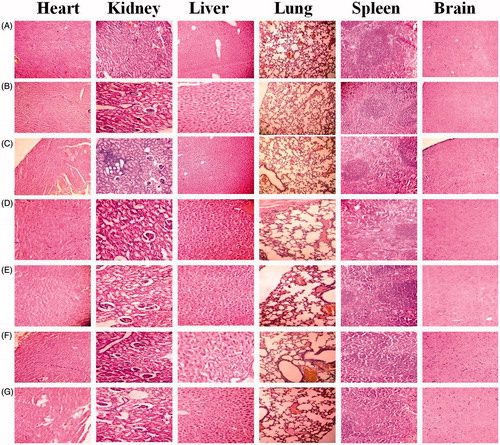
Figure 3. (A) Differential scanning calorimetry thermograms and (B) powder X-ray diffraction crystallographs of (1) Dox, (2) Gantrez® AN 119, (3) PES, (4) NPs, (5) Pul NPs (6) AGn NPs, and (7) Pul–AGn NPs.
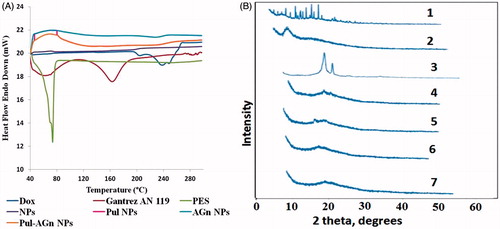
Figure 4. In vitro release of Dox from various Dox formulations (mean ± S.D.; n = 3). (A) Phosphate buffer, pH 7.4. (B) Acetate buffer, pH 5.5.
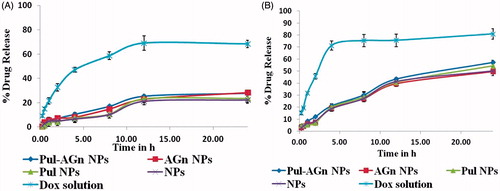
Table 1. Kinetic models for in vitro drug release.
Figure 5. (A) In vitro hemolysis and (B) In vitro serum stability of Dox nanoparticles (mean ± S.D.; n = 3).
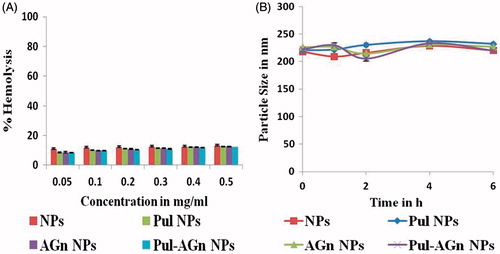
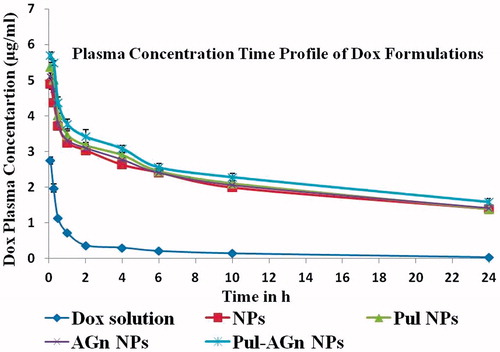
Figure 6. Plasma concentration versus time profiles of Dox following intravenous administration of Dox formulations (mean ± S.D.; n = 6).