Figures & data
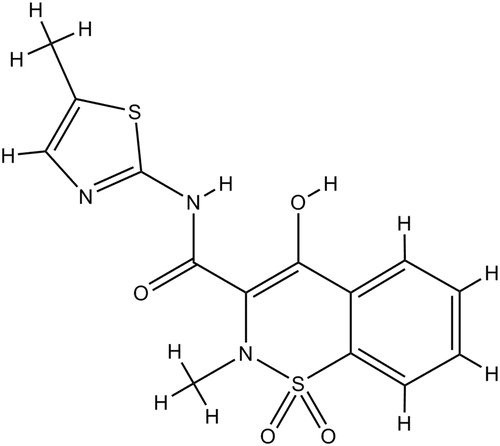
Table 2. Physicochemical properties of idea drug for skin delivery and MXTable Footnotea.
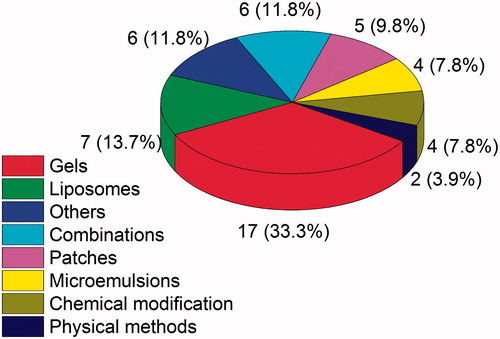
Table 3. Summary of the key findings of studies involving in vitro skin delivery of MX.