Figures & data
Table I. Optimized ratio along with characterization parameters.
Table II. Optimized mannan concentrations along with characterization parameters.
Figure 1. In-vitro drug release of neutral and mannan-coated liposomes of drugs where manna ARIF, AINH, APYZ represent mannan-appended liposomes of RIF, INH, and PYZ.
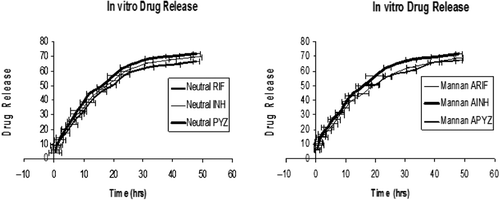
Table III. Percent (%) drug retention (PDR) study.
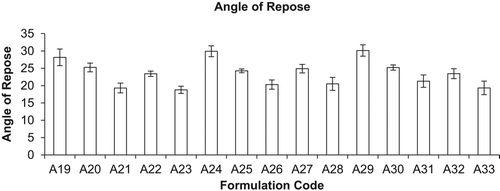
Figure 3. Moisture content (%) of different formulations (mean ± S.D., n = 3). Here A19, A24, and A29 represent inhalation powder of drugs; A20, A25, and A30 represent neutral liposomes of drugs for inhalation; A21, A26, and A31 represent DPI of neutral liposomes of drugs; A22, A27, and A32 represent mannan-coated liposomes of drugs; and A23, A28, and A33 represent DPI of mannan-coated liposomes of drugs, i.e. RIF, INH, and PYZ, respectively.
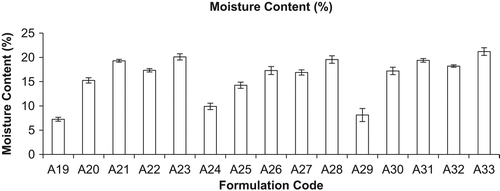
Table IV. Tissue distribution of drugs after administration of different liposomal formulations.