Figures & data
Figure 1. Schematic of multi-probe approach (left) and multi-probes arranged on a robotic end-effector (right). (Three probes are shown as an example: the actual number may vary.)
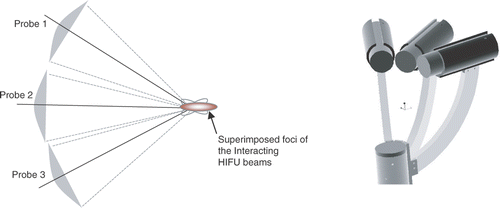
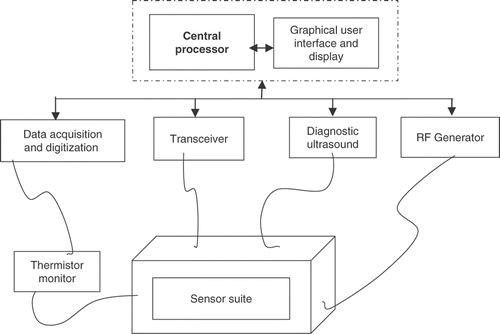
Figure 2. Left: Multiple HIFU transducers mounted on a robotic end-effector submerged in a water tank. Right: The GUI for planning and calibration. [Color version available online.]
![Figure 2. Left: Multiple HIFU transducers mounted on a robotic end-effector submerged in a water tank. Right: The GUI for planning and calibration. [Color version available online.]](/cms/asset/3d3c8dc0-283b-487e-a120-ae5ab31158a1/icsu_a_358850_f0002_b.gif)
Figure 5. Dynamic compensation using lesion position and temperature feedback. [Color version available online.]
![Figure 5. Dynamic compensation using lesion position and temperature feedback. [Color version available online.]](/cms/asset/3775b3b9-8b64-4242-8e2d-a2d9016eecf6/icsu_a_358850_f0005_b.gif)
Figure 9. Color map of the tissue region with respect to the selected noise tolerance. [Color version available online.]
![Figure 9. Color map of the tissue region with respect to the selected noise tolerance. [Color version available online.]](/cms/asset/333c198d-705d-417a-9d82-ac30423af5c4/icsu_a_358850_f0009_b.gif)
Figure 10. Variation of acoustic pulse amplitude with temperature rise (base temperature of the bath = 25°C). [Color version available online.]
![Figure 10. Variation of acoustic pulse amplitude with temperature rise (base temperature of the bath = 25°C). [Color version available online.]](/cms/asset/a598bad8-5891-4091-8a26-56116ce374fb/icsu_a_358850_f0010_b.gif)
Figure 12. (a) The FUSBOT system as seen under the operating table, with target tissue descended through the breast operating window. (b) Lesions induced in porcine kidney with 22 W/8 seconds exposures at 2 mm (top) and 1 mm (bottom) separation. [Color version available online.]
![Figure 12. (a) The FUSBOT system as seen under the operating table, with target tissue descended through the breast operating window. (b) Lesions induced in porcine kidney with 22 W/8 seconds exposures at 2 mm (top) and 1 mm (bottom) separation. [Color version available online.]](/cms/asset/fd3adc9c-619c-4061-aa9e-a7b29f528b56/icsu_a_358850_f0012_b.gif)
Figure 13. Image display on GUI window shown as a result of fusion of sensory data for lesion tracking and thermal mapping. [Color version available online.]
![Figure 13. Image display on GUI window shown as a result of fusion of sensory data for lesion tracking and thermal mapping. [Color version available online.]](/cms/asset/04e2ee00-b00f-48e0-9bca-6ff38a160bef/icsu_a_358850_f0013_b.gif)
Table I. Representative results averaged from 80 experiments in in vitro and ex vivo tissues (porcine adipose and kidneys) for lesion positioning algorithm performance.
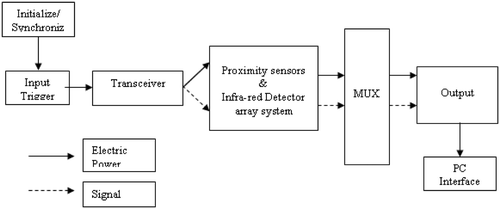
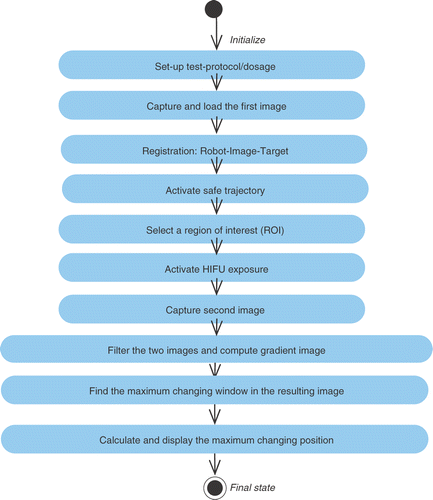
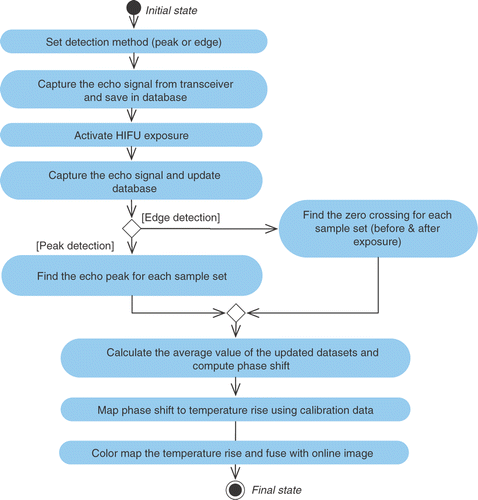
![Figure 8. Image data fusion correlated with temperature map. [Color version available online.]](/cms/asset/a31672d9-972e-4855-8111-9826c23d1e71/icsu_a_358850_f0008_b.gif)
![Figure 11. Phase shift versus temperature. [Color version available online.]](/cms/asset/96e3dc40-6769-4ef0-b427-895cbd242724/icsu_a_358850_f0011_b.gif)
![Figure 14. Gray level versus power for various exposure durations in excised porcine (a) and lamb (b) tissues. [Color version available online.]](/cms/asset/b3ac4fca-6eb0-449a-a300-55f75ea8a136/icsu_a_358850_f0014_b.gif)