Figures & data
Table I. Details of five cases with navigation endoscopic assisted intra-lesional curettage.
Figure 1. CT and MR images for Case 2 were fused in the navigation system and the tumor delineated from the fused image datasets. The tumor volume (outlined in red) can be calculated, enabling an estimate of the amount of bone cement or artificial bone graft required to fill the bone defect after intra-lesional curettage. The locations of working portals were planned and marked by virtual pedicle screws.
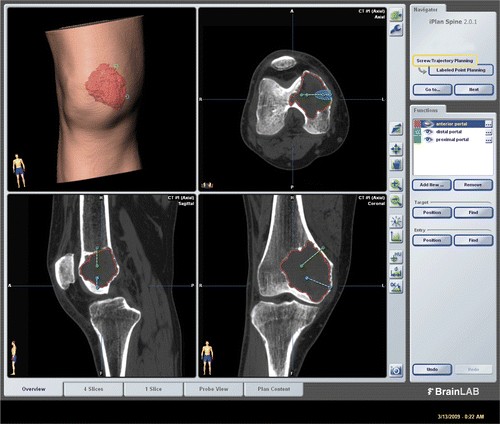
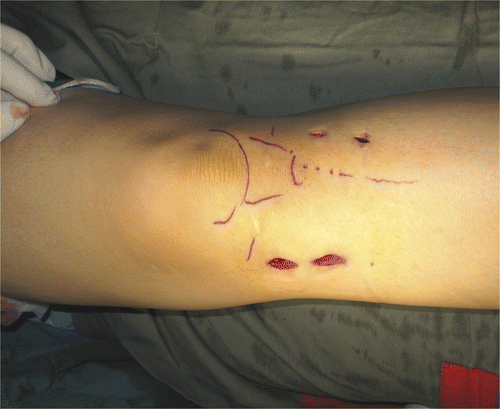
Figure 2. The set-up for NEAT surgery. A shoulder arthroscope and high-speed bone burr mounting a navigation tracker were inserted via two working portals into the tumor cavity. Intra-lesional curettage could be performed under both direct endoscopic visualization and navigation guidance.
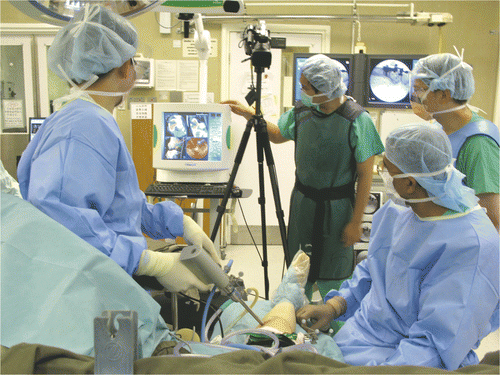
Figure 3. CT-fluoro matching was used for image-to-patient registration in Case 1. This involved the matching of preoperative CT images of a segmented bone region with two intraoperatively acquired fluoroscopic images, thus allowing minimally access registration of the preoperative CT images.
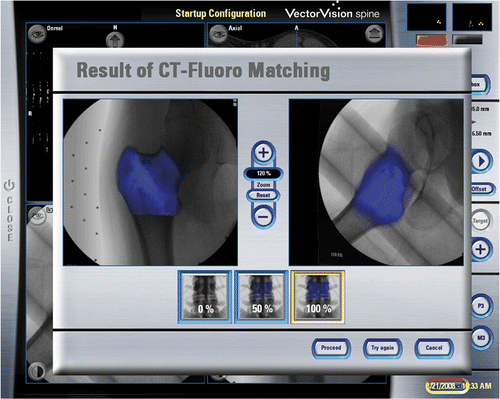
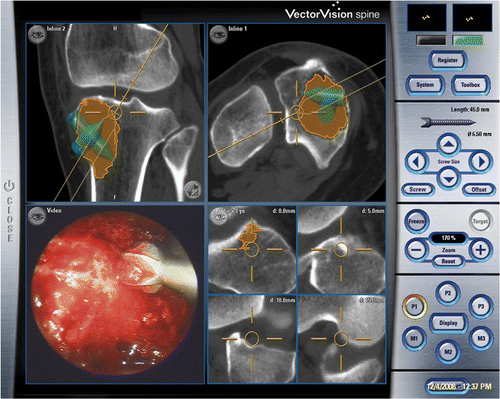
Figure 4. The tumor cavity and bone burring procedure could be directly visualized with the endoscope via small incision portal sites. The location of the navigated instrument tip (the orange circle) is shown with respect to reconstructed CT images. (The tumor is shaded in orange. The extrapolated orange line represents the direction of the instrument and is marked with 5-mm gradations, enabling it to be used to estimate the amount of bone removed and the risk of cortex perforation.)