Figures & data
Figure 1. Structures of pyrazolo[3,4-c]isothiazole and isothiazolo[4,3-d]isoxazole derivatives (5a–g, 6).
![Figure 1. Structures of pyrazolo[3,4-c]isothiazole and isothiazolo[4,3-d]isoxazole derivatives (5a–g, 6).](/cms/asset/de19cacc-62bc-4fa6-9dad-09b106a66520/iphb_a_527350_f0001_b.gif)
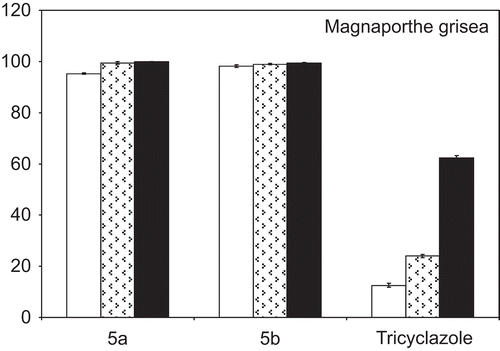
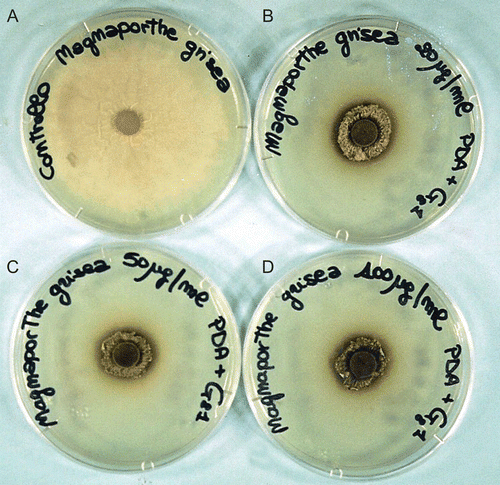
Figure 2. Antifungal activity of the compounds 5a-g and 6 expressed as percentage inhibition of growth on test organisms. The fungi were cultured on PDA in presence of 20, 50, and 100 µg/mL (empty, dotted, and full bars, respectively). Values are means of three trials made in triplicate, Y-error bars indicate standard deviation.
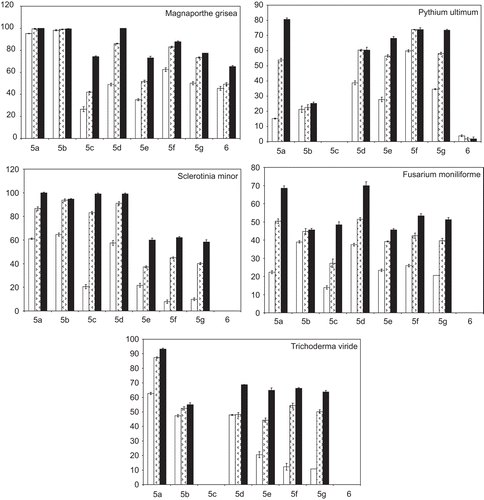
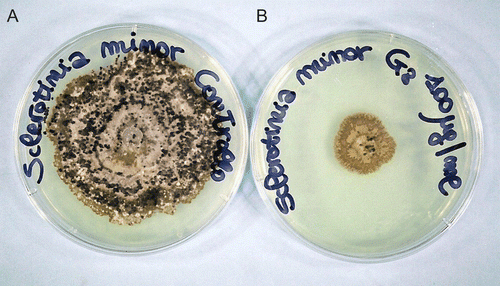
Figure 3. Cultures of Sclerotinia minor control and treated with compound 5a (G8) at 100 µg/mL: in the control are evident black sclerotia, which are instead absent after treatment with 5a (plate on the right).
![Scheme 1. Synthesis of new pyrazolo[3,4-c]isothiazole derivatives.](/cms/asset/fda38ad2-c3c0-4856-81fc-7c200bb6fe78/iphb_a_527350_f0002_b.gif)