Figures & data
Table 1. 1H- and 13C-NMR chemical shifts of AB-1 (3-O-β-d-glucosyl-14-deoxyandrographiside), AB-2 (14-deoxyandrographolide) and AB-3 (14-deoxy-11,12-dihydrondrographolide), (chemical shifts in δ ppm), 600 and 150 MHz in CDCl3, “o” denotes overlapping signals, J values are given in Hertz (Hz).
Table 2. Minimum inhibitory concentration (MIC) and minimum fungicidal concentration (MFC) of DCM and MEOH extracts of A. paniculata whole plant.
Figure 1. (a) Structure of AB-1 (3-O-β-d-glucosyl-14-deoxyandrographiside) obtained from MEOH extract of A. paniculata whole plant.(b) Structure of AB-2 (14-deoxyandrographolide) obtained from MEOH extract of A. paniculata whole plant. (c) Structure of AB-3 (14-deoxy-11,12-didehydrondrographolide) obtained from DCM extract of A. paniculata whole plant.
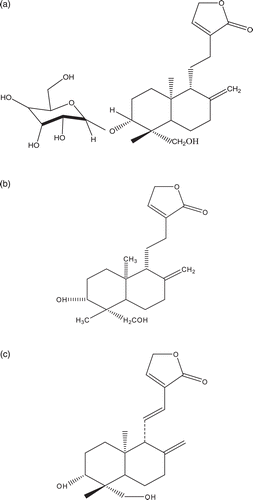
Table 3. Minimum inhibitory concentration (MIC) and minimum fungicidal concentration (MFC) of active principles and positive controls against selected fungal strains.