Figures & data
Figure 1. Chromatogram of EOCO. 1) α-thujene; 2) α-pinene; 3) camphene; 4) β-phellandrene; 5) β-myrcene; 6) α-terpinene; 7) ρ-cymene; 8) limonene; 9) γ-terpinene; 10) α-terpinolene; 11) α-terpineol; 12) bornyl ester; 13) 2-bornyl aetate; 14) α-terpinyl acetate; 15) thujopsene; 16) (+)-δ-cadinene; 17) (+)-epi-bicyclosesquiphellandrene; 18) (+)-ρ-himachalene; 19) (+)-α-longipinene; 20) elemol; 21) α-cedrol; 22) γ-eudesmol; 23) β-eudesmol; 24) beyerene. (Compounds occupying more than 0.4% peak area are shown.)
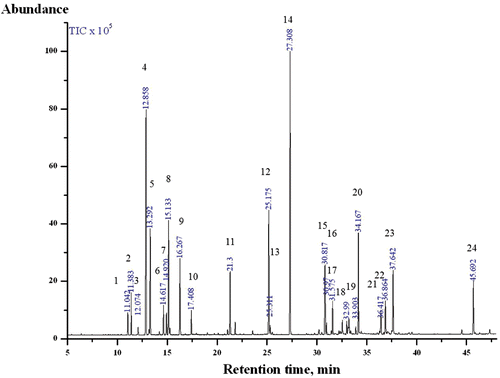
Table 1. Chemical composition of the essential oil extracted from Chamaecyparis obtusa.
Figure 2. Effects of EOCO on Aβ1–40-induced impairments of memory acquisition and retention in the Morris water maze test. Before receiving intrahippocampally injection of either ACSF or Aβ1–40 (4 μL), the rats inhaled EOCO (1 mL/cage) in a VSC for 30 days. Donepezil (1 mg/kg b.w./day) served as a positive control. The rats were given four trials each day for 4 consecutive days. (A) The effects of EOCO on the memory of rats impaired by injection of Aβ1–40 in the fourth day of the Morris water maze test. (B) The effects of EOCO on the memory of rats impaired by injection of Aβ1–40 in the first trial and fourth trials of the Morris water maze test over 4 days. The values shown are the mean latency ± S.D (n = 7); *p < 0.05, **p < 0.01, ***p < 0.001, significantly different from the Aβ1–40-treated group. Statistical significance was tested with the unpaired Student’s t-test.
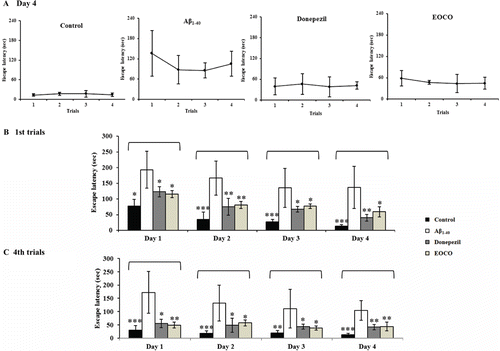
Table 2. Effects of EOCO on Aβ1–40-induced impairment of memory acquisition and memory retention in the passive avoidance test.
Figure 3. Effects of EOCO on Aβ1–40-induced impairments of memory acquisition and retention in probe trials. (A) Effects of EOCO on probe trial sessions of the Morris water maze test. The time in the figure represents the cumulative time in the target quadrant of the pool in the 90 s probe trial. (B) Swimming pattern on probe trial sessions of the Morris water maze test. The values shown are the mean latency ± S.D (n = 7); *p < 0.05, **p < 0.01, ***p < 0.001, significantly different from Aβ1–40-treated group. Statistical significance was tested with the unpaired Student’s t-test.
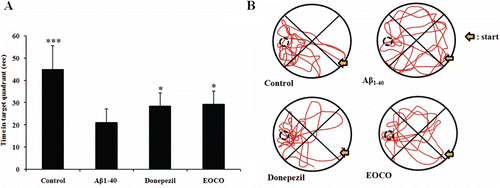
Figure 4. Effects of EOCO on Aβ1–40-induced behavioral deficits. (A) Effects of EOCO on Aβ1–40-induced sensorimotor deficits in the vertical pole test. (B) Effects of EOCO on Aβ1–40-induced motor coordination and balance deficits in the rotarod test. The values shown are the mean latency ± S.E. (n = 7); *p < 0.05, **p < 0.01, ***p < 0.001, significantly different from Aβ1–40-treated group. Statistical significance was tested with the unpaired Student’s t-test.
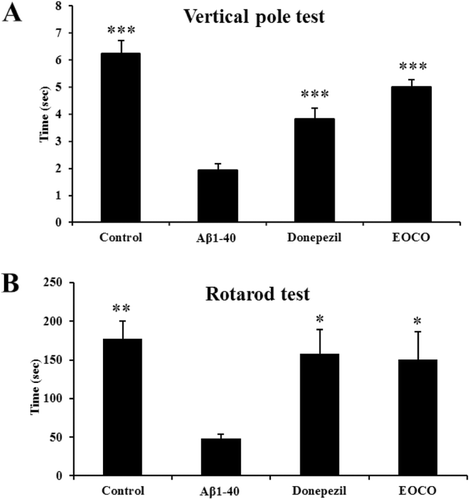
Figure 5. Effects of EOCO on AchE activity (A) and ACh content (B) in Aβ1–40-injured rats. Results are expressed as the means ± S.D. (n = 7); *p < 0.05, **p < 0.01, ***p < 0.001, significantly different from only Aβ1–40-treated group. Statistical significance was tested with the unpaired Student’s t-test.
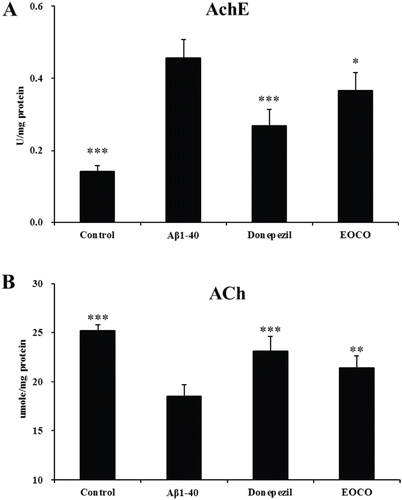
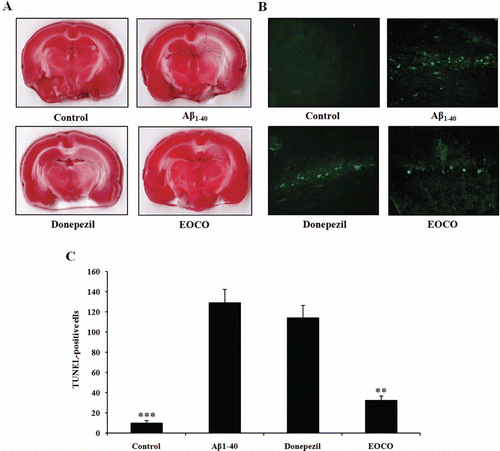
Figure 6. Effects of EOCO on Aβ1–40-induced neuronal degeneration and DNA strand breakage. (A) Effects of EOCO on Aβ1–40-induced neuronal degeneration assessed by TTC staining. Representative brain sections (2 mm thick) from EOCO-treated rats stained with 2% TTC showing neuronal degeneration. Red colored regions in the TTC-stained sections indicate nondegenerated regions and pale colored regions indicate degeneration. (B) Effects of EOCO on Aβ1–40-induced neuronal apoptosis assessed by TUNEL assay. Detection of hippocampal apoptotic cells was carried out using the TUNEL method. TUNEL-positive pyramidal neurons undergoing apoptosis were observed in the hippocampus (fluorescence). (C) Representative quantification of the number of TUNEL-positive cells. Results are expressed as the means ± S.E. (n = 7); *p < 0.05, **p < 0.01,***p < 0.001, significantly different from Aβ1–40-treated group. Statistical significance was tested with the unpaired Student’s t-test.