Figures & data
Table 1. Proximate composition in the fruiting bodies of G. lucidum.
Table 2. Comparison of composition in different fractions and the sacchachitins obtained by different processes from the fruiting bodies of G. lucidum.
Table 3. Assignment of IR absorption for different products and their deacetylation degree.
Figure 2. The IR spectra of sacchachitins and sacchachitosans. (A) Our product sacchachitn and MSC (the mimic CitationSu et al., 1997). (B) Our product sacchachitosan and (C) sacchachitosan from MSC. The characteristic absorption reads in (A) (at cm–1): νO–H 3455.7–3450.5, νN–H 3500–3300, νC–H 2932.5–2932.4, 2300–2400 unassigned, νC=O 1736.3–1734.6, amide I δN–H 1551.4–1550.4, δC–H 1413.5–1324.5, νC–C 1154.6–1152.7, νC–N 1125.9–1121.7, νC–O 1029.5–1028.7, δN–H 780.2–779.5, δC–H 713.3–712.3, and δC=O 690.5-690.2. Similar absorption was seen in (B) and (C), but lacking the bands νC=O 1736.3–1734.6 and the amide I bands δN–H 1551.4–1550.4.
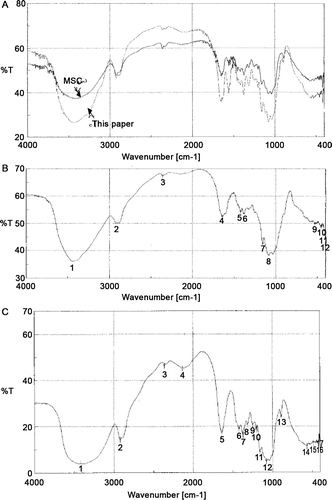
Table 4. %T of characteristic IR absorption bands before and after deacetylation.
Table 5. Pattern of monosaccharide composition in different fractions obtained from the fruiting bodies of G. lucidum.
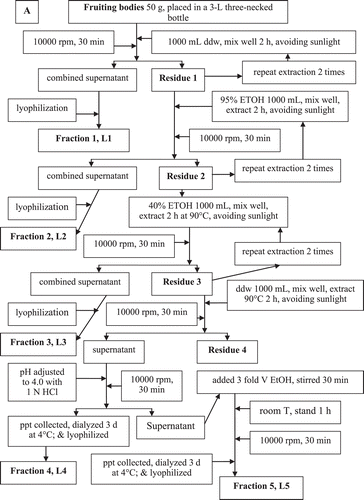
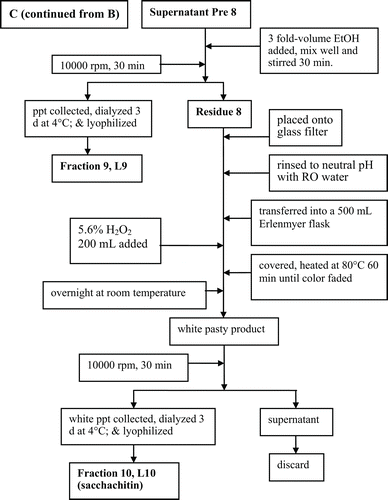
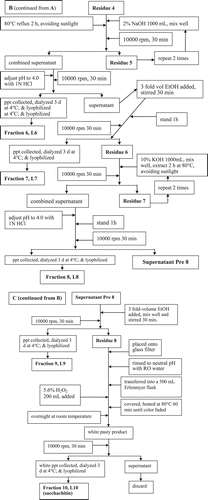
Figure 3. The scavenging capability of different polysaccharide fractions of G. lucidum on DPPH radicals (A), the superoxide anions (B), and the hydroxyl free radicals (C). Values are expressed in means ± SD (n = 3). Fractions L5–L10 are indicated in the flow chart in .
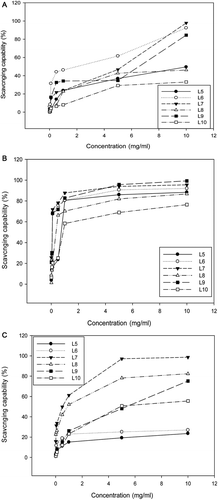
Figure 4. Scavenging capability of the two different sacchachitins isolated by different processes from the fruiting bodies of G. lucidum. For the DPPH radicals (A); for the superoxide anions (B); and for the hydroxyl free radicals (C). Values are expressed in means ± SD (n = 3).
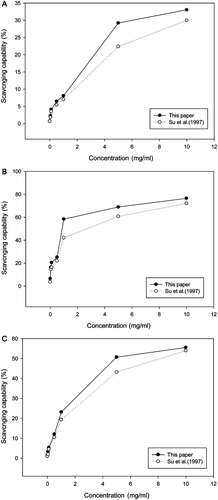
Table 6. Antioxidative IC50 values of different polysaccharide fractions obtained from the fruiting bodies of G. lucidum.