Figures & data
Figure 1. Typical chromatograms of LIG from rat plasma: (A) blank plasma sample; (B) blank plasma sample spiked with LIG; (C) plasma sample obtained at 1 h from rat after an i.v. administration of LIG (12.5 mg/kg); (D) standard solution of LIG spiked with IS. Peak 1, podophyllotoxin (internal standard); Peak 2, LIG.
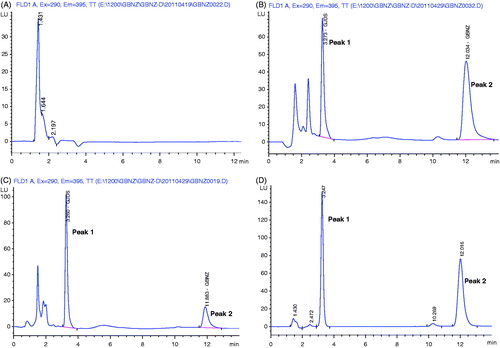
Table 1. Precision and accuracy data for LIG in rat plasma (n = 5).
Table 2. Extraction recoveries of LIG from rat plasma (n = 5).
Table 3. Summary of stability studies of LIG in rat plasma under various storage conditions (n = 5).
Figure 2. Plasma concentration--time curves of LIG in rats after oral and intravenous administration (n = 6). (A): intravenous administration of 12.5, 25 and 50 mg/kg, b.w. LIG; (B): oral administration of 12.5, 25 and 50 mg/kg, b.w. LIG.
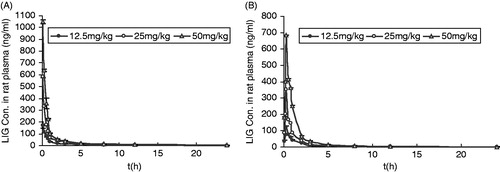
Figure 3. Relationship between Cmax and AUC vs. dose in rats receiving single 12.5, 25 and 50 mg/kg, b.w. intravenous dose of LIG.
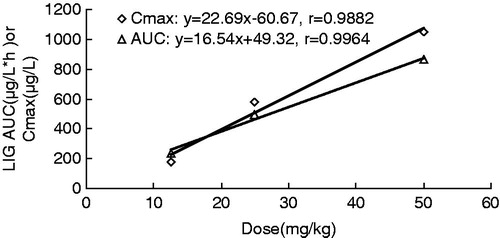
Table 4. Pharmacokinetic parameters of LIG in rats after oral and intravenous administration at three different doses (n = 6, mean ± SD).
