Figures & data
Figure 1. Study protocol for experimental groups and timing of SB203580 (SB) or normal saline solution (NSS) administration.
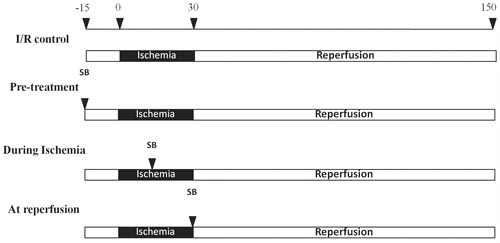
Figure 2. Effects of SB203580 on cardiac mitochondrial function. (A) Western blot analysis using antibody against mitochondrial VDAC1/Porin in cytoplasmic fraction (C), and mitochondrial fraction (M) of isolated cardiac mitochondria. (B) Mitochondrial swelling, (C) mitochondrial ROS production, (D) mitochondrial membrane potential change, when SB203580 was administered before, during ischemia, or at the onset of reperfusion in ischemia/reperfusion rats (n = 4–7 animals/group). *p < 0.05 versus the vehicle group, #p < 0.05 versus the pretreatment group.
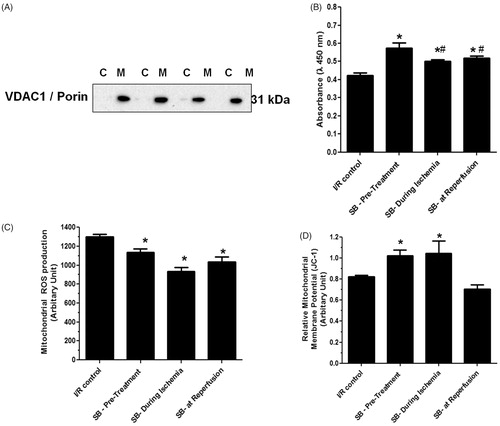
Figure 3. Effects of SB203580 on cardiac mitochondrial ultrastructure. Cardiac mitochondria were isolated from rats subjected to myocardial ischemia/reperfusion injury (A) in the presence of SB203580 before (B), during ischemia (C), or at the onset of reperfusion (D). The mitochondrial pellet was embedded in EM-bed 812 resin. Ultrathin sections were cut and stained with uranyl acetate and lead citrate. *p < 0.05 versus the vehicle group (n = 4–5 animals/group).
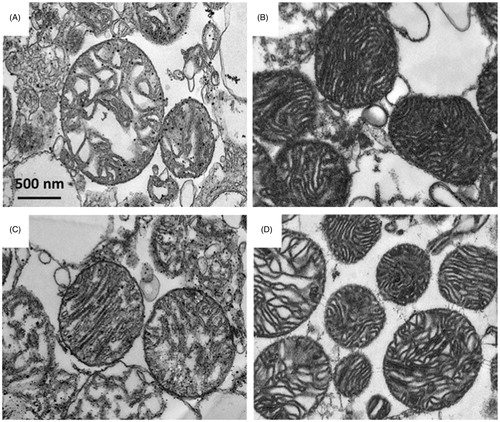
Figure 4. (A) Effects of SB203580 on p38 MAPK activation and downstream substrates, HSP27, p53, and CREB. The heart homogenates were collected and subjected to Western blot analysis to detect the activation of interested proteins. (B–E) The quantitation of fold phosphorylation of p38 MAPK activation and downstream substrates, HSP27, p53, and CREB by Western blot analysis (n = 3 animals/group). *p < 0.05 versus the vehicle group.
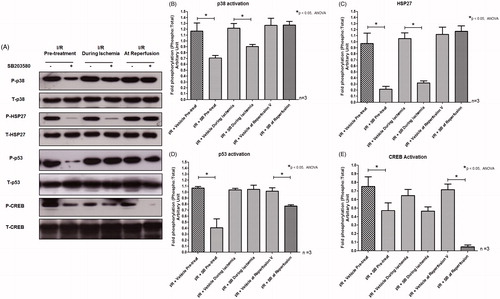
Figure 5. (A) Effects of SB203580 on apoptotic regulatory proteins. The heart homogenates were collected and subjected to Western blot analysis to detect the expression of Bax, Bcl2, Cytochrome c, and cleaved caspase-3. (B–E) The quantitation of protein expression of Bax, Bcl2, Cytochrome c, and cleaved caspase-3 by Western blot analysis (n = 3 animals/group). *p < 0.05 versus the vehicle group.
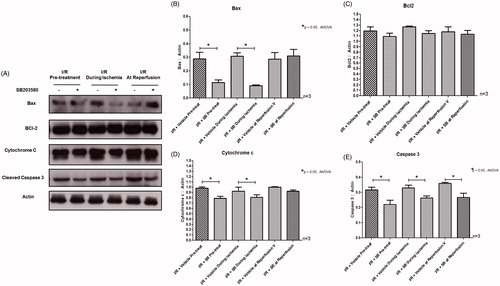
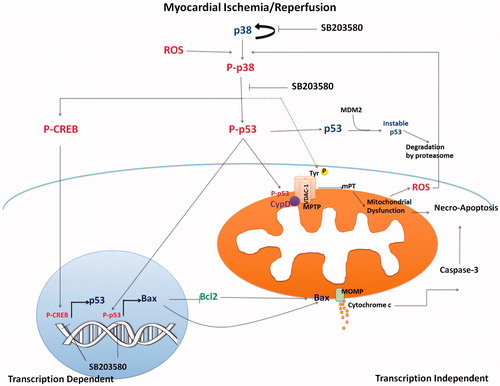
Figure 6. The proposed mechanistic pathway of I/R-activated p38 MAPK and downstream activation involving mitochondrial trigger cell death. Myocardial I/R injury caused p38 MAPK activation, which consequently activated p53 phosphorylation. Phosphorylated p53 stabilized and accumulated in mitochondrial matrix during I/R injury and mediated MPTP opening. Activation of p53 also activated proapoptotic Bax protein, which regulated cytochrome c release, and activation of caspase 3 (transcription independent manner). p38 MAPK also phosphorylated CREB, which in turn regulated p53 gene transcription. Inhibition of p38 MAPK by SB203580 reduced p53 phosphorylation, CREB phosphorylation, and thus protected cardiac mitochondria from I/R injury and cell death.