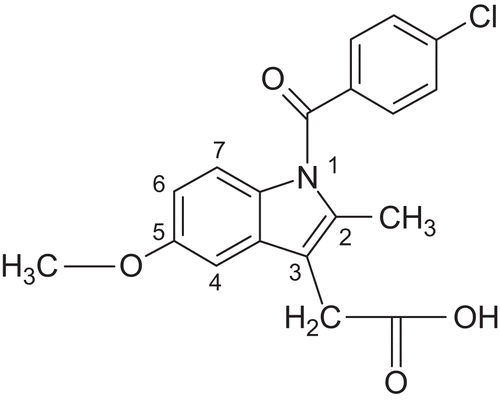
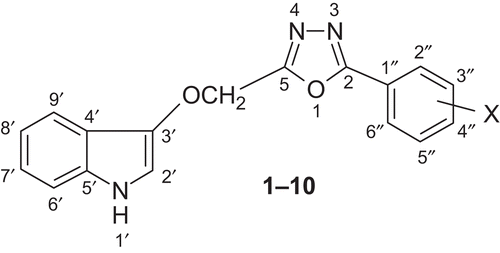
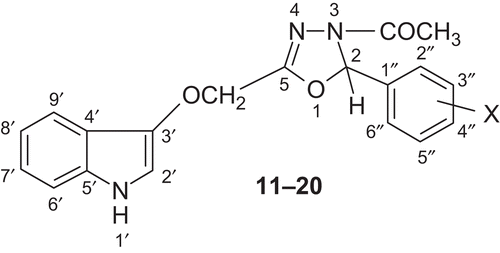
Figures & data
Scheme 1. Synthesis of oxadiazole and oxadiazoline analogs. Reagents and conditions: (a) ClCH2COOC2H5, K2CO3, acetone, reflux for 12 h; (b) NH2NH2, ethanol, reflux for 8 h; (c) aldehydes, ethanol, reflux for 6 h; (d) Br, CH3COONa, CH3COOH, 2 h stirring; (e) (CH3CO)2O, reflux for 1 h.
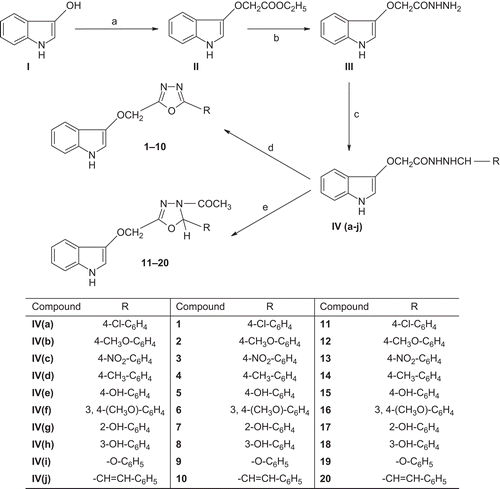
Table 1. Anti-inflammatory activitya of the synthesized oxadiazoles and oxadiazolines (100 mg/kg p.o.) using carrageenan-induced rat paw edema method and cotton pellet-induced granuloma method.
Table 2. Ulcerogenicity index of the synthesized oxadiazole and oxadiazoline analogs (20 mg/kg i.p.).