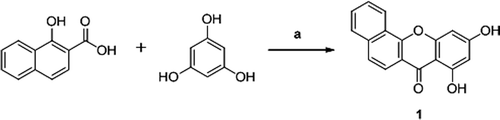
Figures & data
Scheme 2. Reagents and conditions: (a) Br2 (2 eq), AcOH, r.t.; (b) NIS, DMF, r.t.; (c) Br2 (3 eq), AcOH, r.t.; (d) 2-hydroxybenzyl alcohol, ZnCl2, 100°C, dioxane; (e) different acyl chloride, Et3N, acetone, r.t.; (f) benzyl bromide, K2CO3, acetone, ref.; (g) corresponding acyl chloride, NaH, THF (dry), r.t.; (h) 3-methoxybenzoyl chloride, Et3N, acetone, r.t., then 3-bromobenzoyl chloride, NaH, THF (dry), r.t.
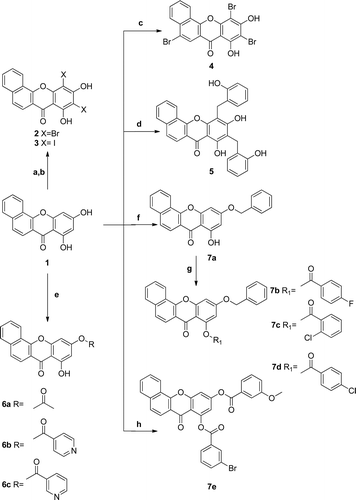
Table 1. The IC50 (μM) values of the synthesized compounds against three tumour cell lines.
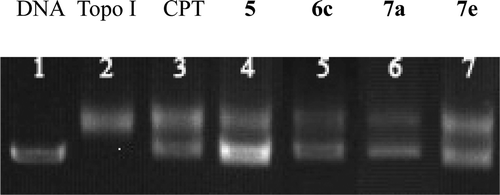
Figure 1. Effect of the selected compounds on topoisomerase-I-mediated DNA relaxation in single concentration. Topo I was Topoisomerase I and DNA without drugs. The samples were reacted with 100 μM drugs at 37°C for 15 min. Reactions were then stopped by adding 0.5% SDS. Gels were photographed under UV transilluminator.