Figures & data
Scheme 1. Reagents and conditions: (a) for compound 3: compound 2, MeO–NH2·HCl, pyridine, room temperature to 80 °C, 1 h, 70%; (b) for compound 4: compound 2, H2NC(=O)–NH–NH2·HCl, Na-acetate, ethanol–water, room temperature, overnight, 60%.
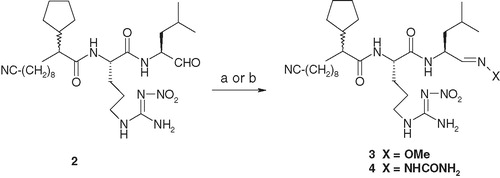
Table 1. Biological data for compounds.*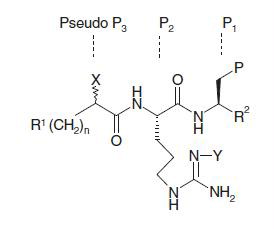 .
.
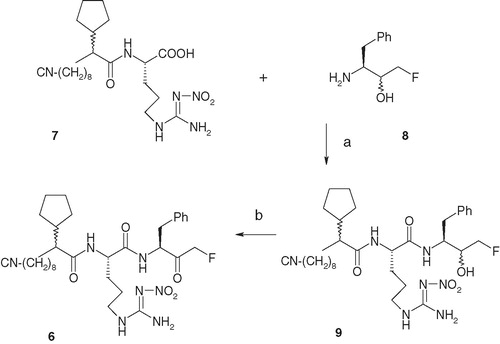
Scheme 2. Reagents and conditions: (a) BOP, HOBt, NMM and DMF, 0 °C to room temperature, 2 h, 60%; (b) Dess–Martin reagent, CH2Cl2, 0 °C to room temperature, 2 h, 70%.
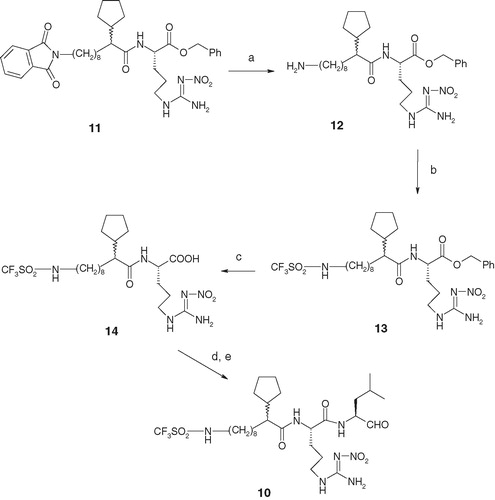
Scheme 3. Reagents and conditions: (a) hydrazine monohydrate, MeOH, heat, 1 h; (b) CF3SO2Cl, Et3N, CH2Cl2, −10 °C to room temperature, 2 h, 60% over two steps; (c) H2 gas, catalytic 10% Pd–C (50% water content), MeOH, 42–26 psi, 2 h, 80%; (d) (S)-leucinol, BOP, HOBt, NMM, DMF, 0 °C to room temperature, 2 h, 70%; and (e) Dess–Martin reagent, CH2Cl2, 0 °C to room temperature, 2 h, 80%.