Figures & data
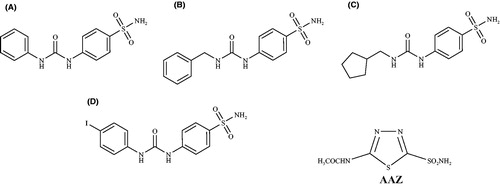
Scheme 1. Preparation of sulfonamides 6–20 by reaction of 4-amino-N-(4-sulfamoyl-benzyl)-butyramide hydrochloride (n = 1) (4) or 4-amino-N-[2-(4-sulfamoyl-phenyl)-ethyl]-butyramide hydrochloride (n = 2) (5) with arylisocyanates A1–8 (R-NCO) in presence of diisopropylethylamine (DIPEA), in acetonitrile (dry).
![Scheme 1. Preparation of sulfonamides 6–20 by reaction of 4-amino-N-(4-sulfamoyl-benzyl)-butyramide hydrochloride (n = 1) (4) or 4-amino-N-[2-(4-sulfamoyl-phenyl)-ethyl]-butyramide hydrochloride (n = 2) (5) with arylisocyanates A1–8 (R-NCO) in presence of diisopropylethylamine (DIPEA), in acetonitrile (dry).](/cms/asset/891c4214-cda6-412c-ae2e-17363cd7b997/ienz_a_1014477_sch0001_b.jpg)
Table 1. Isocyanates used for the preparation of compounds 6–20.
Table 2. Inhibition data of human CA isoforms hCA I, II, IX and XII with ureido-sulfonamides 6–20 reported here and the standard sulfonamide inhibitor acetazolamide (AAZ) by a stopped flow CO2 hydrase assayCitation26.  .
.
Figure 1. Graphical comparison of inhibition constants of the new ureido-sulfonamides 6–20 reported here and the standard drug AAZ against hCA I, II, IX and XII.
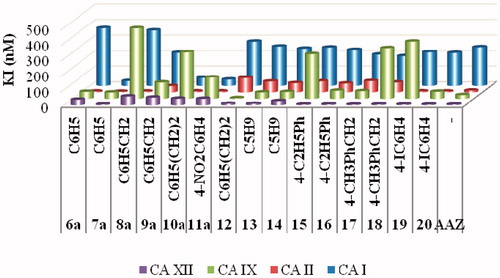
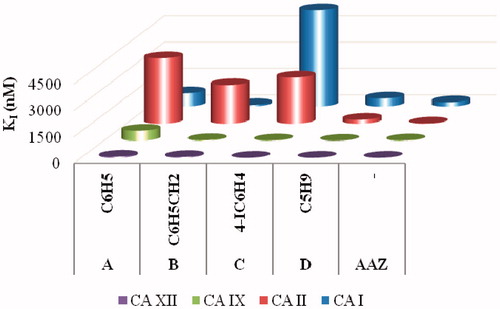
Figure 2. Graphical comparison of inhibition constants of the ureido-sulfonamides A–D previously reported and the standard drug AAZ against hCA I, II, IX and XII.