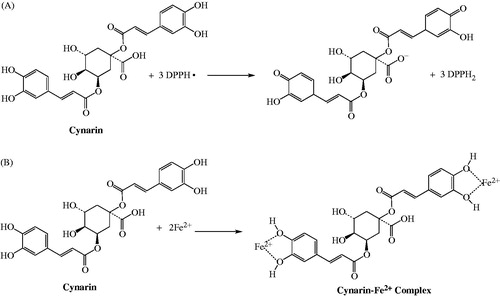
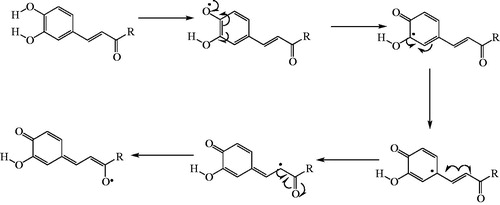
Figures & data
Figure 1. Total antioxidant activities of cynarin and standard antioxidant compounds like trolox, α-tocopherol, BHT, and BHA at the same concentration (30 µg/mL) assayed by the ferric thiocyanate method. The control value reached a maximum 50 h (BHA, butylated hydroxyanisole; BHT, butylated hydroxytoluene).
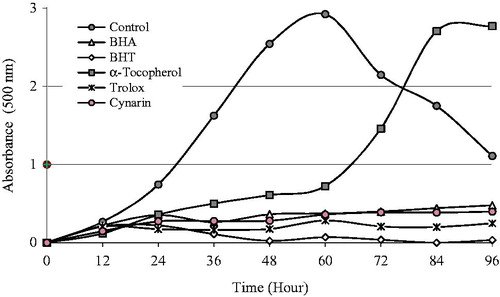
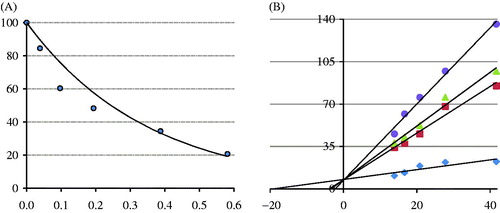
Figure 2. Reducing power of cynarin. (A) Fe3+ → Fe2+ reductive potential of different concentrations (10–30 µg/mL) of cynarin (r2: 0.983) and reference antioxidants. (B) Cu2+ reducing ability of different concentrations (10–30 µg/mL) of cynarin (r2: 0.840) and reference antioxidants (BHA, butylated hydroxyanisole; BHT, butylated hydroxytoluene).
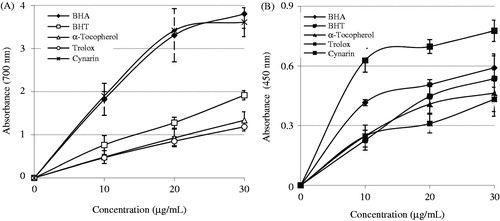
Figure 3. Comparison of Fe2+ chelating activity of cynarin (r2: 0.898) and standard antioxidant compounds like trolox, α-tocopherol, BHT, and BHA at the concentrations of 10–20 µg/mL (BHA, butylated hydroxyanisole; BHT, butylated hydroxytoluene).
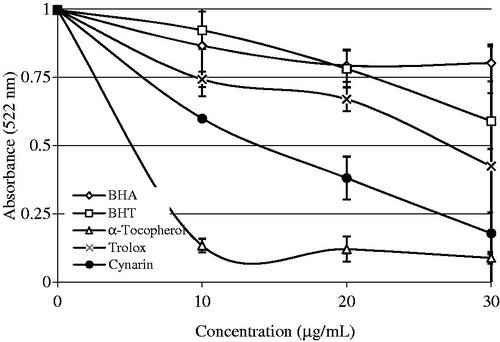
Figure 4. Comparison of superoxide anion radical () scavenging activities of cynarin and standard antioxidant compounds like trolox, α-tocopherol, BHT, and BHA at the concentration of 30 µg/mL (BHA, butylated hydroxyanisole; BHT, butylated hydroxytoluene).
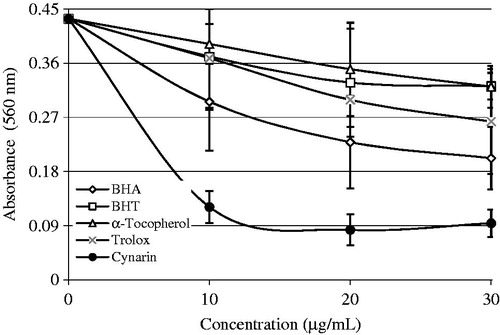
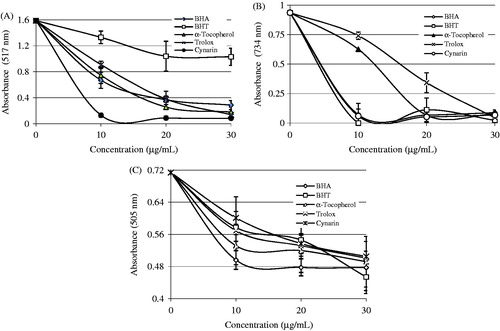
Figure 5. Radical-scavenging activities of cynarin. (A) DPPH free radical scavenging activity of different concentrations (10–30 µg/mL) of cynarin (r2: 0.950) and reference antioxidants. (B) ABTS radical scavenging activity of different concentrations (10–20 µg/mL) of cynarin (r2: 0.956) and reference antioxidants. (C) DMPD radical scavenging activity of different concentrations (10–30 µg/mL) of cynarin (r2: 0.982) and reference antioxidants (BHA, butylated hydroxyanisole; BHT, butylated hydroxytoluene; DPPH•, 1,1-diphenyl-2-picryl-hydrazyl free radical; ABTS•+, 2,2′-Azino-bis(3-ethylbenzthiazoline-6-sulfonic acid; DMPD•+, N,N-dimethyl-p-phenylenediamine radical).