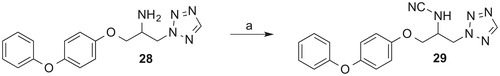
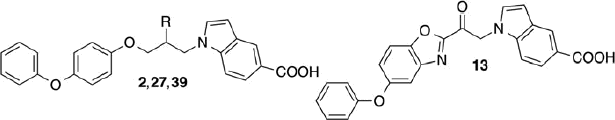
Figures & data
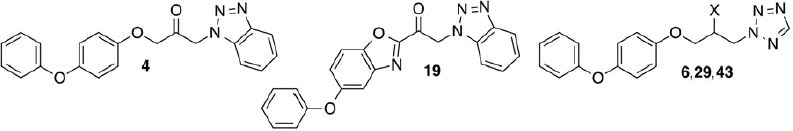
Figure 1. Heteroaryl-substituted propan-2-ones with preferential inhibition of cPLA2α (1 and 2) and FAAH (3–6).
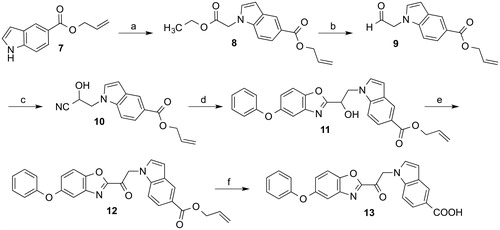
Scheme 1. Reagents and conditions: (a) Ethyl bromoacetate, Cs2CO3, KI, acetonitrile, room temperature, 16 h; (b) diisobutylaluminium hydride, cyclohexane, THF, −70 °C to room temperature, 4 h; (c) acetone cyanhydrin, triethylamine, CH2Cl2, room temperature, 4 h; (d) 1. acetyl chloride, ethanol, chloroform, 0 °C, 1 h; 2. 2–amino-4-phenoxyphenol, ethanol, reflux, 6 h; (e) Dess-Martin periodinane, CH2Cl2, room temperature, 4 h; (f) tetrakis(triphenylphosphine)palladium(0), acetic acid, THF, room temperature.
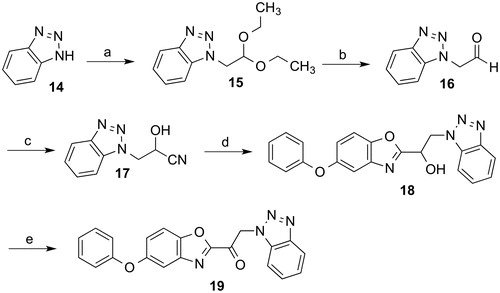
Scheme 2. Reagents and conditions: (a) Bromoacetaldehyde diethyl acetal, DMF, NaHCO3, reflux, 18 h; (b) conc. HCl, THF, room temperature, 5 h; (c) acetone cyanhydrin, triethylamine, CH2Cl2, room temperature, 5 h; (d) 1. acetyl chloride, ethanol, chloroform, 0 °C, 1 h; 2. 2–amino-4-phenoxyphenol, ethanol, reflux, 6 h; (e) Dess-Martin periodinane, CH2Cl2, room temperature, 4 h.
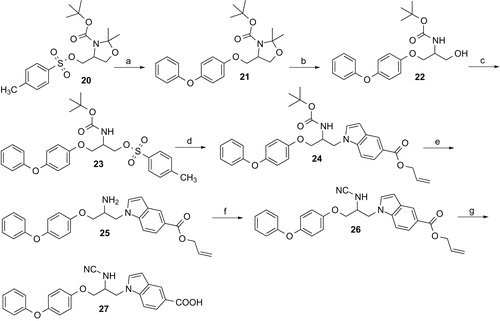
Scheme 3. Reagents and conditions: (a) 4-Phenoxyphenol, NaH, DMF, 70 °C, 3 h; (b) p-toluenesulfonic acid, methanol, room temperature, 4 h; (c) tosyl chloride, 4-dimethylaminopyridine, triethylamine, CH2Cl2, room temperature, 12 h; (d) allyl indole-5-carboxylate, NaH, DMF, 80 °C, 3 h; (e) trifluoroacetic acid, CH2Cl2, room temperature, 2 h; (f) cyanogen bromide, CH2Cl2, NaHCO3, THF, 0 °C, 2 h; (g) tetrakis(triphenylphosphine)palladium(0), acetic acid, THF, room temperature.
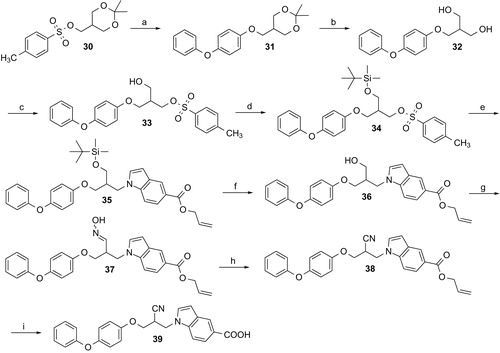
Scheme 5. Reagents and conditions: (a) 4-Phenoxyphenol, NaH, DMF, 80 °C, 3 h; (b) 5 M HCl, methanol, room temperature, overnight; (c) p-toluenesulfonyl chloride, 4-dimethylaminopyridine, triethylamine, THF, room temperature, 12 h; (d) tert–butyldimethylsilyl chloride, CH2Cl2, triethylamine, room temperature, 4 h; (e) allyl indole-5-carboxylate, NaH, DMF, 70 °C, 3 h; (f) tetrabutylammonium fluoride, THF, room temperature, 1 h; (g) 1. Dess-Martin periodinane, acetic acid, CH2Cl2, room temperature, 1.5 h; 2. hydroxylammonium chloride, pyridine, room temperature, overnight; (h) 2-chloro-1-methylpyridinium iodide, THF, triethylamine, room temperature, 20 h; (i) tetrakis(triphenylphosphine)palladium(0), acetic acid, THF, room temperature.
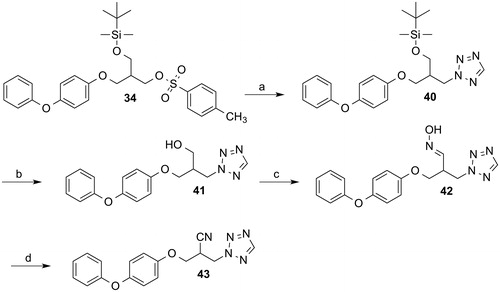
Scheme 6. Reagents and conditions: (a) Tetrazole, acetonitrile, DMF, NaOH, 100 °C, 2 h; (b) tetrabutylammonium fluoride, THF, room temperature, 2 h; (c) 1. Dess-Martin periodinane, acetic acid, CH2Cl2, room temperature, 1.5 h; 2. hydroxylammonium chloride, pyridine, room temperature, overnight; (d) 2-chlor-1-methylpyridinium iodide, THF, triethylamine, room temperature, 20 h.