Figures & data
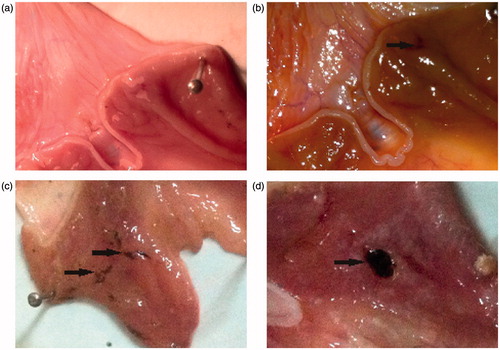
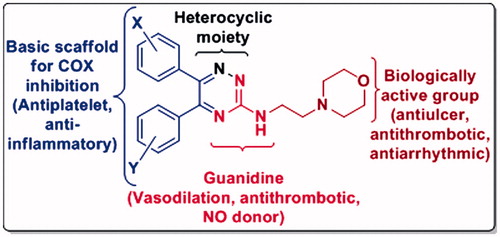
Figure 2. Designing hypothesis for the new compounds by fusing three pharmacophores in a single molecule for antiplatelet and vasodilatory action without ulcerogenesis.
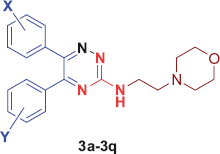
Scheme 1. Green synthesis of 5,6-diaryl-N-morpholinoethyl-1,2,4-triazin-3-amines (3a–3q). Reagents and conditions: (a) thiosemicarbazide, methyl iodide, ionic liquid [Bbim+Br−]: DMSO (1:10), 70 °C, 10–110 min; (b) 4-(2-aminoethyl)morpholine (4 eq.), neat, 100–110 °C, 2–4 h.
![Scheme 1. Green synthesis of 5,6-diaryl-N-morpholinoethyl-1,2,4-triazin-3-amines (3a–3q). Reagents and conditions: (a) thiosemicarbazide, methyl iodide, ionic liquid [Bbim+Br−]: DMSO (1:10), 70 °C, 10–110 min; (b) 4-(2-aminoethyl)morpholine (4 eq.), neat, 100–110 °C, 2–4 h.](/cms/asset/98563f35-53c6-497f-bfb5-5789834ddd55/ienz_a_1060480_sch0001_c.jpg)

 .
.