Figures & data
Figure 1. Schematic representation of oxidative stress. (a) In a healthy individual, there is an abundant of antioxidant capacities in the lungs to counteract inhaled oxidants from air pollution or acute exposures to cigarette smoke. (b) However, chronic smokers experience a reduced antioxidants and increased oxidant burden delivered from the cigarettes. At an early stage, the oxidant burden is still compensated by the antioxidant capacity but eventually (c) the oxidant burden overwhelms the body's antioxidant protection mechanism, leading to oxidative stress and the development of COPD.
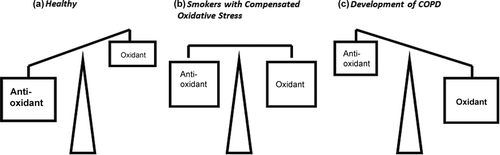
Table 1. Endogenous and exogenous sources of oxidants
Figure 2. Consequences of airway damage by oxidative stress. Oxidative stress causes airway damages at three levels: airway changes, cellular changes, and biochemical changes. These factors contribute to lung injury and inflammation, which leads to the development of COPD. The ongoing oxidant injuries and inflammation in COPD feeds back to the increased airway damage, restarting the vicious cycle.
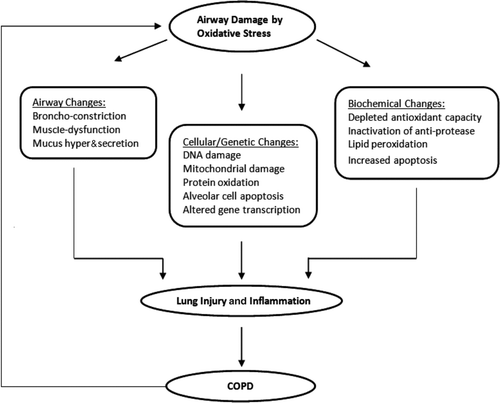
Figure 3. Vicious cycle of oxidative stress and inflammation (adapted from Lee 2009) (154). Oxidative stress generated by cigarette smoking increases ROS production in the bronchial mucosa, leading to the recruitment of inflammatory cells, which further increases oxidative stress. These phenomena cause lung injuries, which further enhances lung tissue inflammation and the generation of more ROS.
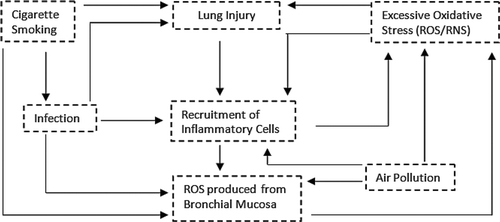
Table 2. Mechanisms of controlled apoptosis (Recreated from Demedts 2006; Hodge 2005) (30, 35)
Table 3. Summary of genetic association studies of COPD with EcSOD (Reproduced from Oberley-Deegan et al. 2009) (78)
Figure 4. Diagrammatic representation of the GSH redox system. The oxidant hydrogen peroxide (H2O2) generated from oxidative stress is neutralized by the glutathione redox system with the assistance of GSH-reductase and glutathione peroxidase.
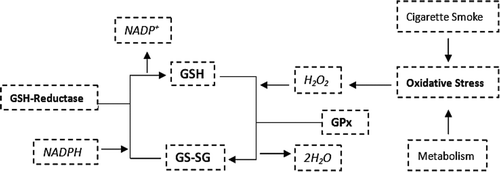
Figure 5. Integrated diagram of oxidative stress and the heme breakdown pathway by HO-1. Heme is a product of haemoglobin metabolism and is further broken down into carbon monoxide, iron, and biliverdin, which is then converted into bilirubin. Each of these downstream products of heme metabolism has antioxidant capacities that function at different levels. CO acts as anti-inflammatory, anti-apoptotic, anti-proliferative agent. The iron-storage protein ferritin has antioxidant actions that counteract oxidative stress, and bilirubin is also an antioxidant and anti-inflammatory molecule.
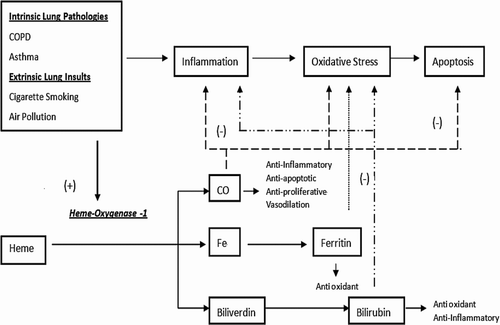
Table 4. Recent clinical trials for the efficacy of antioxidants in pulmonary disease and in the general population