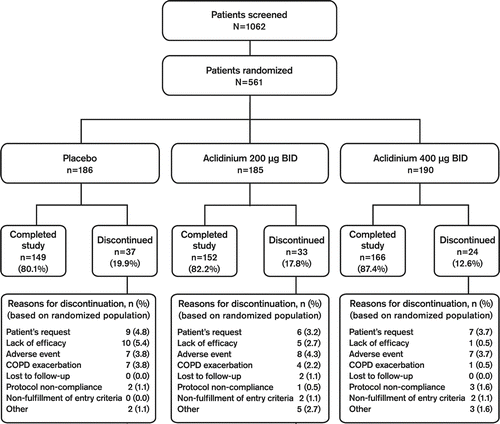
Figures & data
Table 1. Demographic data and baseline characteristics (safety population)
Figure 2. Mean (SE) change from baseline in (A) trough FEV1 and (B) peak FEV1 at Day 1 (peak only) and at Weeks 1, 4, 8 and 12.
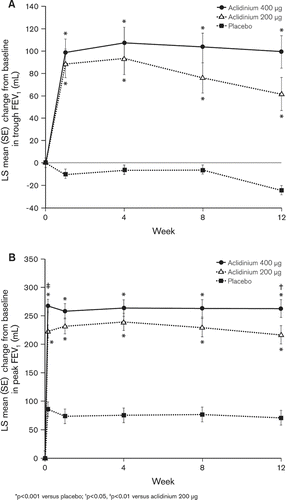
Table 2. Mean (SE) change from baseline in pulmonary function parameters (FVC and IC) after 12 weeks of treatment (ITT population)
Figure 3. Mean (SE) change from baseline in A) SGRQ total score and B) TDI focal score at Weeks 4, 8, and 12.
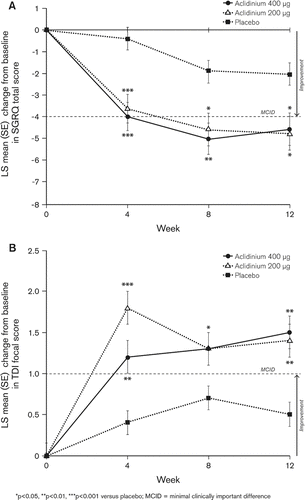
Figure 4. Percentage of patients who achieved a clinically meaningful difference in (A) SGRQ total score (≥4 units) and (B) TDI focal score at Weeks 4, 8 and 12.
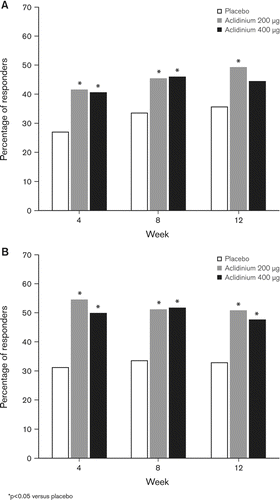
Table 3. Mean (SD) change from baseline in daily average of COPD nighttime and early morning symptom scores at Week 12 (ITT population)
Table 4. Most frequently reported (≥2% of subjects in any group) adverse events by treatment group (n [%]; safety population; N = 560)