Figures & data
Figure 1. Study design. Following an initial screening phase, subjects were enrolled into this crossover study, which involved two treatment periods (1 and 2), each consisting of two treatments (60 or 120 mg/kg weekly IV alpha1-PI (Prolastin-C®), separated by a washout period. Time points for serial samples for pharmacokinetic (PK) analysis are indicated.
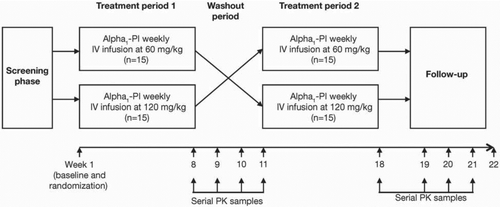
Figure 2. Subject flow. aSubject had previously received gene therapy for alpha1-PI in a research study.
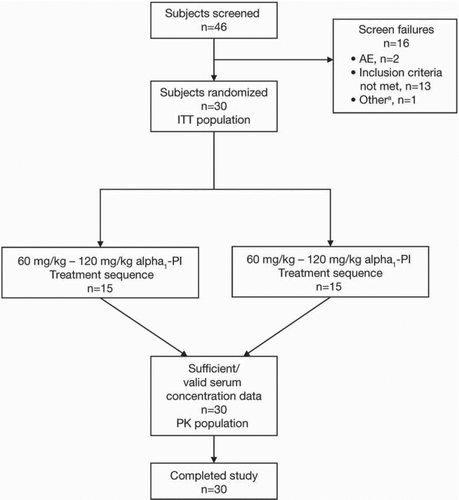
Table 1. Demographics and disease characteristics of the study population
Table 2. Adverse events
Figure 3. Mean steady-state serum alpha1-PI concentration versus time curves. Serum concentrations of alpha1-PI following the eighth weekly IV dose of 60 mg/kg and 120 mg/kg of alpha1-PI were measured in blood samples. Linear scale (A); semi-logarithmic scale (B). aAt week 8, after receiving the 60 mg/kg dose, serum alpha1-PI concentration hardly increased from the pre-dose level throughout the entire PK blood sampling period in one subject. This subject was excluded from PK data analysis.
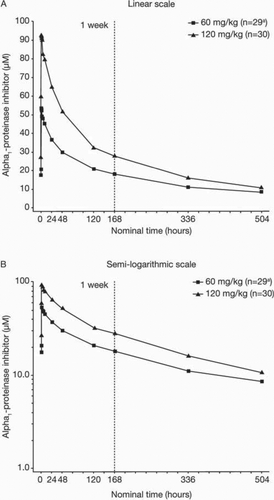
Table 3. Summary of steady-state PK parameters of serum alpha1-PI following weekly intravenous infusion dose of 60 mg/kg and 120 mg/kg of alpha1-PI
Table 4. Statistical analysis of dose-normalized PK parameters of serum alpha1-PI at steady-state following weekly intravenous infusion dose of 60 mg/kg and 120 mg/kg of alpha1-PI