Figures & data
Table 1. A questionnaire was filled in by the subject after nebulization.
Table 2. Baseline characteristics of enrolled subjects.
Figure 1. (A) the change in FEV1 before-after nebulization in stable COPD patients. (STONAC 1). Change in FEV1 was measured at each nebulization. No clinical relevant reduction of FEV1 (> 20%) occurs after nebulizations of amoxicillin clavulanic acid. (B) the change in FEV1 before-after nebulization in COPD exacerbation patients. (STONAC 2). Change in FEV1 was measured once for each subject. No clinical relevant reduction of FEV1 (> 20%) occurs after nebulizations of amoxicillin clavulanic acid.
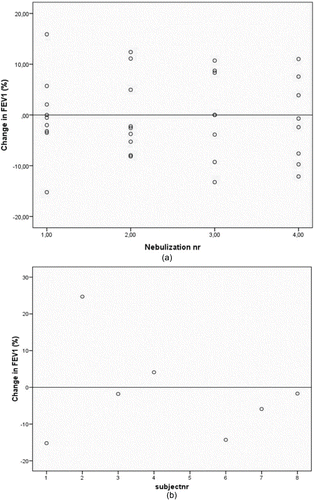
Figure 2. STONAC 1 Amoxicillin concentration in sputum samples collected within 3 hours after nebulization.
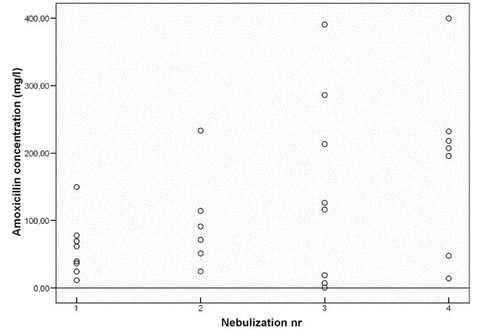
Table 3. STONAC 2: Amoxicillin concentration in sputum samples collected before the second nebulization (trough samples)
