Figures & data
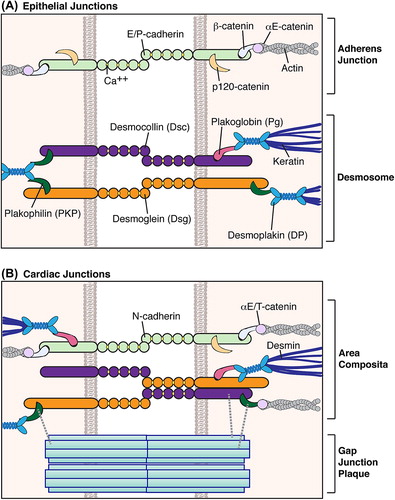
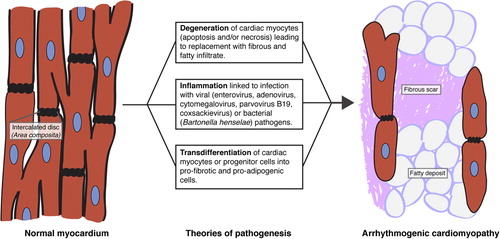
Table 3. Potential mechanisms of AC mutation pathogenicity. Select findings from mouse models and/or in vitro studies are highlighted here, with further information available in the text.