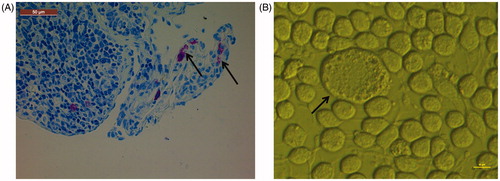
Figures & data
Table 1. Main characteristics of an ideal cell model.
Table 2. Summary of cell models for allergenicity evaluation and clinical diagnosis.