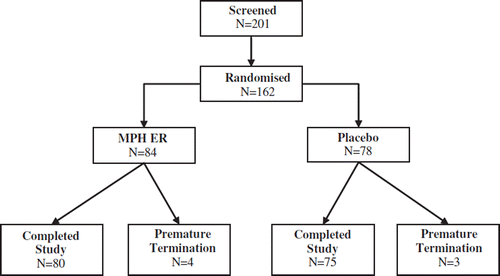
Figures & data
Table I. Demographic and clinical characteristics of the intent-to-treat study population at screening. Data are presented as N (%) or mean ± SD.
Table II. Probable and confirmed DSM-IV axis-1 and axis-II diagnoses according to SCID-I and -II interviews of the intent-to-treat study population (N).
Figure 2. Mean reduction of the WRAADDS total score at week 8 by treatment group (ITT population, Wilcoxon U-test, P = 0.0003).

Figure 3. Mean reduction of the CAARRS-S:L total score at week 8 by treatment group (ITT population, Wilcoxon U-test, P = 0.008).

Figure 4. Mean reduction of the SDS total score at week 8 by treatment group (ITT population, Wilcoxon U-test, P = 0.017).

Figure 5. Responders according to reduction of the WRAADDS total score by 30% in the MPH ER and the placebo group.
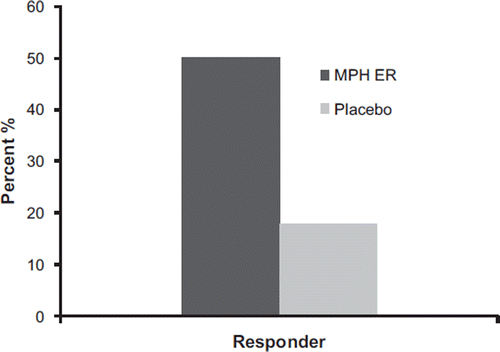
Table III. Adverse events measured by somatic symptom scale of AMDP. Only those adverse events are listed, which were significantly more frequent in either treatment group.
Table IV. Week by week pulse (bpm), systolic and diastolic blood pressure (BP) values. P values are given for treatment group differences.