Figures & data
Table 1. Transcriptomic analysis of oocytes across several species.
Figure 1. Overpopulated GO Process Networks for A) genes differentially expressed in models of developmental competency in oocytes and B) genes differentially expressed in models of developmental competence in cumulus cells.
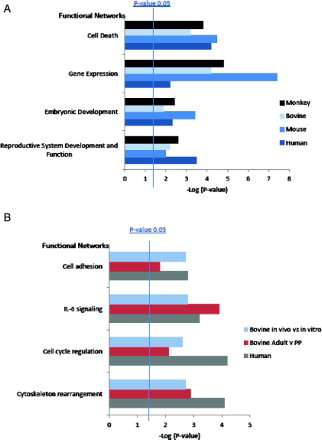
Table 2. Microarray studies performed on models of developmental competence in several species in both oocytes and cumulus cells.
Figure 2. Venn diagramrepresentation of A) the transcriptome of GV stage bovine oocytes [Mamo et al. Citation2011] compared to in vivo matured and in vitro matured bovine oocytes [Kues et al. Citation2008] and B) oocytes matured in vitro and in vivo in bovine [Kues et al. Citation2008] and Monkey [Lee et al. Citation2008].
![Figure 2. Venn diagramrepresentation of A) the transcriptome of GV stage bovine oocytes [Mamo et al. Citation2011] compared to in vivo matured and in vitro matured bovine oocytes [Kues et al. Citation2008] and B) oocytes matured in vitro and in vivo in bovine [Kues et al. Citation2008] and Monkey [Lee et al. Citation2008].](/cms/asset/916e27ce-2d26-44ae-bc08-d9a7a10929bf/iaan_a_656217_f0002_b.jpg)
Table 3. Transcripts upregulated in oocytes from TFF patients and bovine GV oocytes.
Table 4. Transcripts associated with increased developmental competence in oocytes in two or more datasets.
Table 5. Transcripts associated with decreased developmental competence in oocytes in two or more datasets.
Table 6. Transcripts associated with either increased or decreased competence in cumulus cells from several models of developmental competence.
Table 7. Transcription regulation of transcripts differentially expressed in oocytes.
Table 8. Transcription regulation of transcripts differentially expressed in cumulus cells. Datasets from different models of developmental competence were used; bovine in vivo vs. in vitro, human early vs. late cleaved blastocysts, and bovine adult vs. prepubertal MII oocytes.
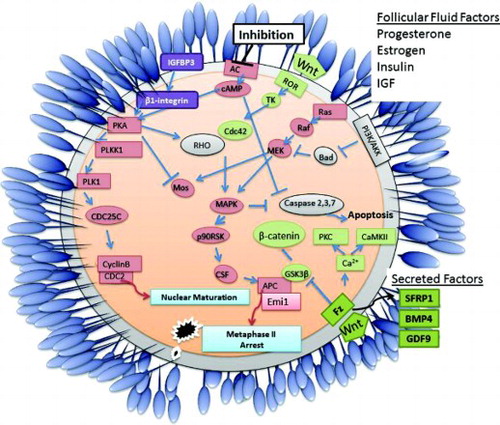
Figure 3. Schematic diagram detailing primary events and pathways associated with acquisition of competence in mature vertebrate oocytes. Oocyte maturation is initiated following extracellular inhibition of adenylate cyclase (AC) which induces the translation of specific mRNAs (such as mos) leading to the activation of maturation promoting factor (cyclinB/cdc2). The activity of Oocyte maturation, Wnt signaling and the PI3K/Akt apoptosis pathways are key to the maturation of a high quality oocyte. Factors produced by the cumulus cells and/or components of the follicular fluid such as progesterone, estrogen, insulin, and IGF may impact on oocyte development and quality. Secreted factors such as BMP4, GDF9, and SFRP1 are potential non-invasive biomarkers of oocyte quality.
TK: tyrosine kinase, Fz: frizzled receptor