Figures & data
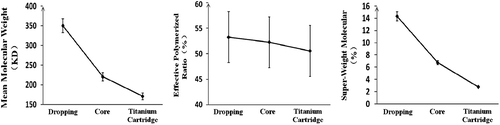
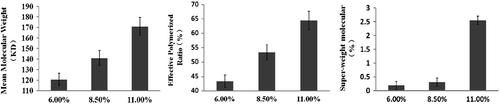
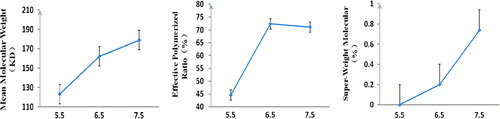
Table I. The molecular weight distribution of poly-hemoglobin and final products based on optimal process (n = 3).
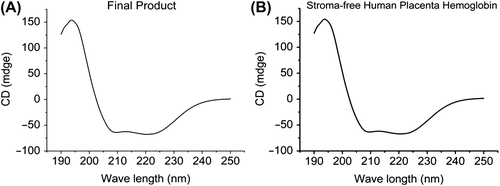
Figure 5. Secondary structure of final products with circular dichroism (A). Compared with the secondary structure of the storma-free human placenta hemoglobin (B), the secondary structure of final products is consistent. This can be very obviously explained by the process of crosslinking without disrupting the secondary structure.