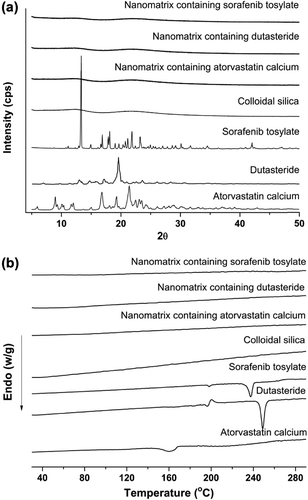
Figures & data
Figure 1. SEM of Aerosil 200 (a) and Soluplus-coated colloidal silica nanomatrix (SCCSN) containing atorvastatin calcium (b), dutasteride (c), and sorafenib tosylate (d).

Figure 3. Kinetic solubility of raw material, physical mixture, and SCCSN for atorvastatin calcium (a), dutasteride (b), and sorafenib tosylate (c) (data represented as mean ± SD, n = 3).
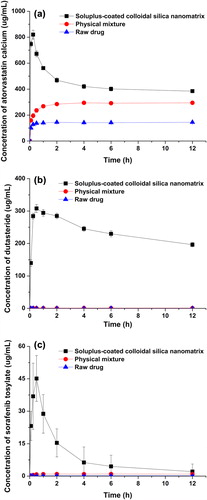
Figure 4. Plasma concentration–time profile of dutasteride in rats (data represented as mean ± SD, n = 4).
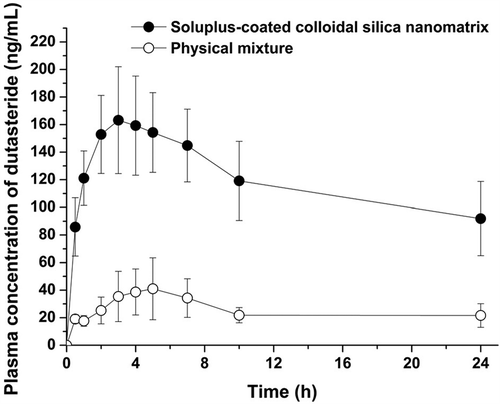
Table I. Pharmacokinetic parameters of dutasteride in rats (data represented as mean ± SD, n = 4).