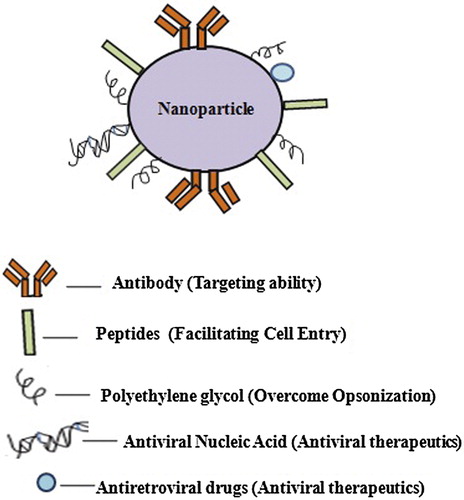
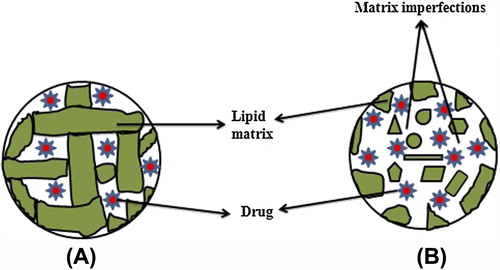
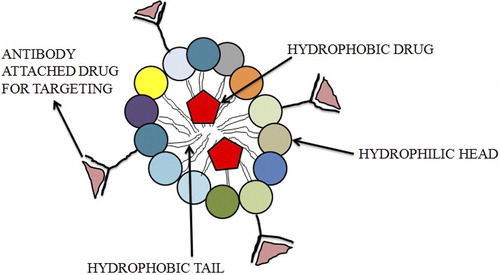
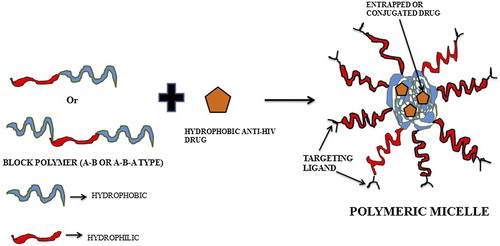
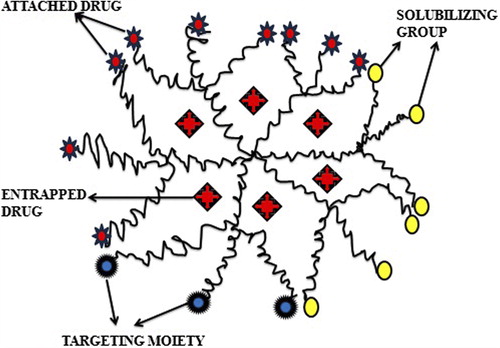
Figures & data
Table I. Marketed anti-retroviral drugs.
Table II. Bioavailability of some dosage form of antiretroviral drugs.
Table III. Fixed dose combination for anti-HIV therapy.
Table IV. Physicochemical and biopharmaceutical characteristics of anti-HIV drugs.
Table V. Advantages and limitations of nanosystems used in HIV-AIDS treatment.
Table VI. Brief summary of different nanotechnology-based systems of potential antiretroviral drugs.
Table VII. List of patents associated with nanosystems used in HIV-AIDS treatment.