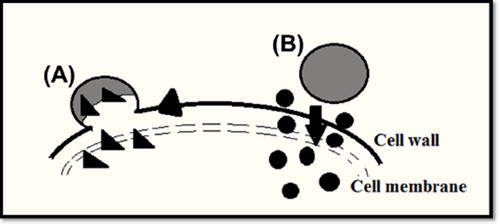
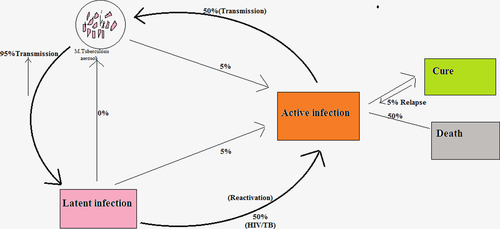
Figures & data
Table I. Species of Mycobacteria that cause tuberculosis.
Table II. Rate of tuberculosis cases by race/ethnicity in United States.
Table III. Classification of antitubercular drugs.
Table IV. Side effects of Anti TB drugs.
Table V. Salient features of drug carriers.
Table VI. Outcomes of liposome-based drug delivery systems in the treatment of TB.
Table VII. Outcomes of nanoparticle-based drug delivery systems in the treatment of TB.
Table VIII. Outcomes of microparticle/microsphere-based drug delivery systems in the treatment of TB.
Table IX. Outcomes of solid lipid nanoparticle based drug delivery systems in the treatment of TB.