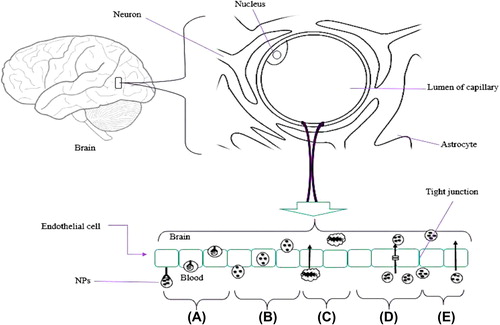
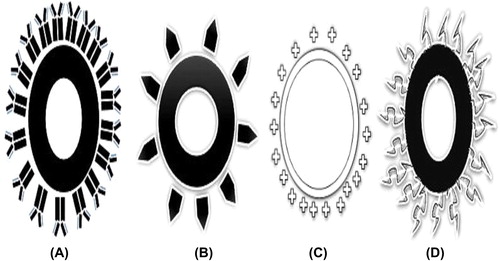
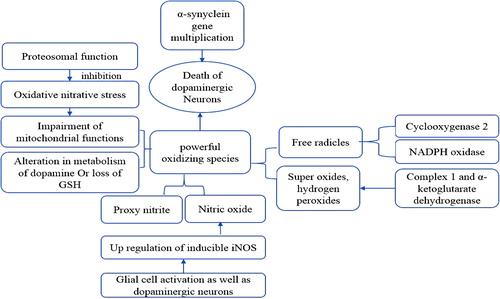
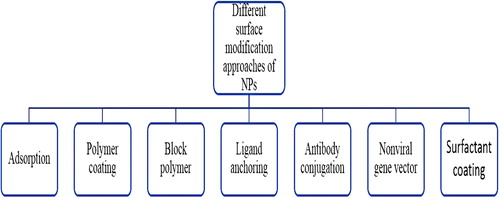
Figures & data
Table II. Various pharmaceutical approaches to overcome the BBB.
Table III. Various studies on surface-modified nanoparticles and their potential to overcome.