Figures & data
Table I. Targets for CAR-modified T cells.
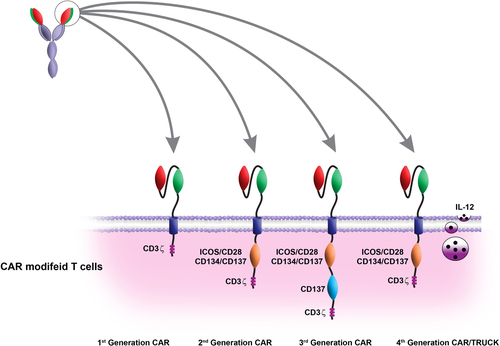
Table II. Different characteristics of the four generations of CAR T cells.
Table III. Clinical trials with different types of CAR-modified T cells.