Figures & data
Figure 1. Synthesis of hydrophobically modified Bletilla striata polysaccharides with different fatty acids (m: the amount of carbon).
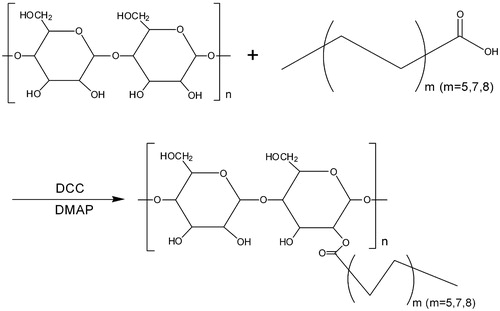
Figure 2. FT-IR spectra of BSP and its derivatives (a: BSP, b: hm-BSP-C12, c: hm-BSP-C16, d: hm-BSP-C18).
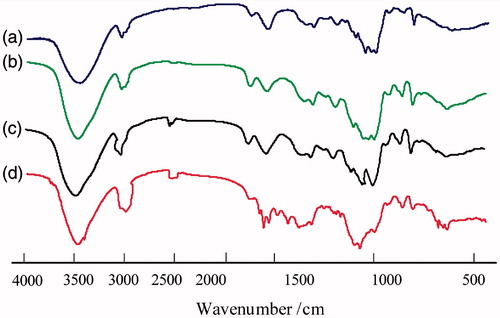
Figure 3. The 1H NMR spectra of BSP and its derivatives (a: BSP, b: hm-BSP-C12, c: hm-BSP-C16, d: hm-BSP-C18) in deuterated DMSO.
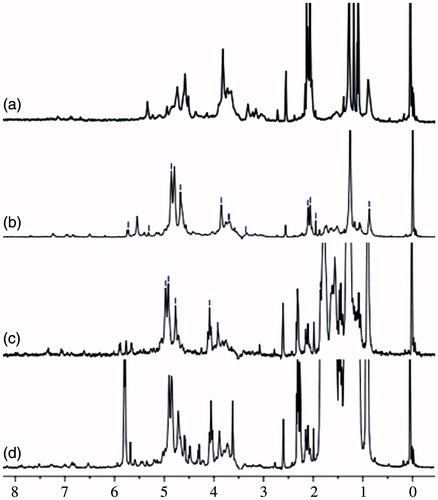
Table 1. Properties of hm-BSPs.
Figure 4. Change of the intensity ratio (I373/I390) from emission spectra of pyrene (6.0 × 10−7) with various concentrations of hm-BSPs in distilled water.
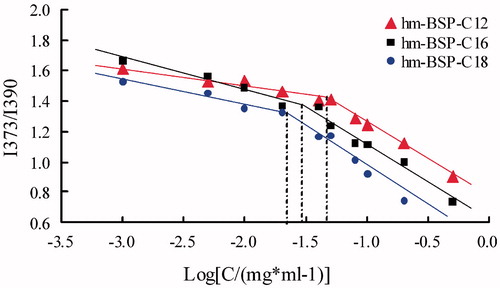
Figure 5. Appearance for BSP and hm-BSP solutions (a: hm-BSP-C12, b: hm-BSP-C16, c: hm-BSP-C18, d: BSP).