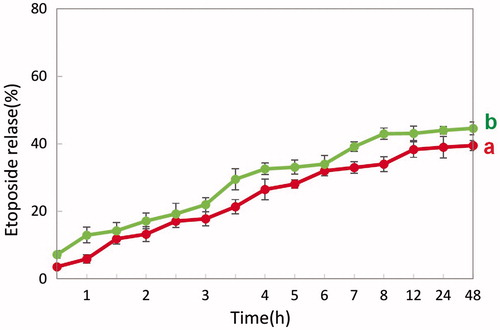
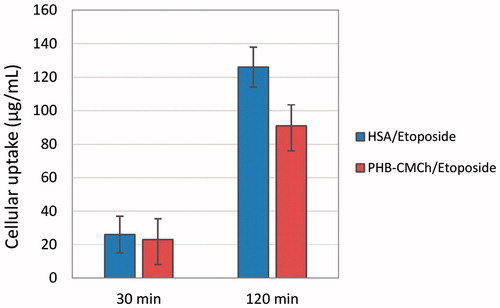
Figures & data
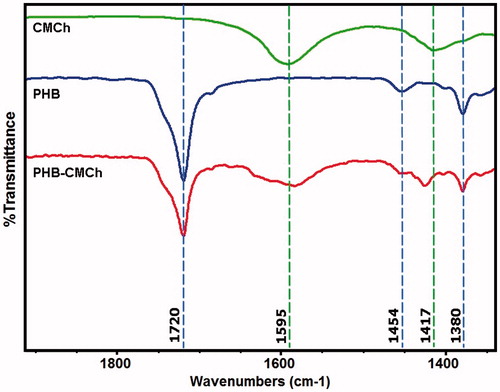
Figure 1. (A) Preparation steps of HSA nanoparticles, B(I) synthesis of o-carboxymethyl chitosan (CMCh), B(II) preparation steps of PHB–CMCh nanoparticles.
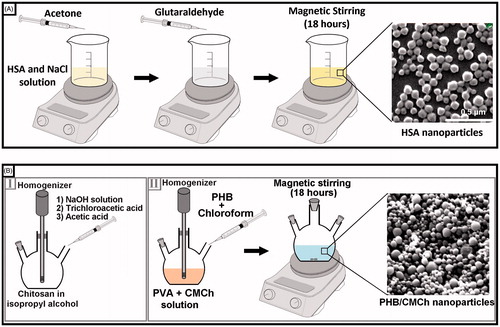
Table 1. Effect of the pH value, degree of crosslinking, polymer concentration on the size of HAS nanoparticles.
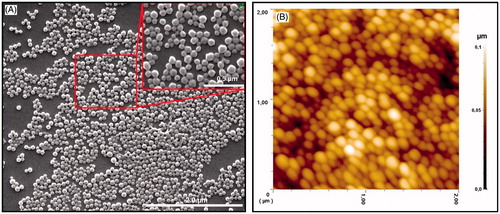
Figure 4. (A) SEM image of the PHB–CMCh nanoparticles and (B) 2D AFM image of the PHB–CMCh nanoparticles.

Table 2. Effect of the concentration of surfactant, polymer and stirring rate on the size, zeta potential, and polydispersity of PHB–CMCh nanoparticles.
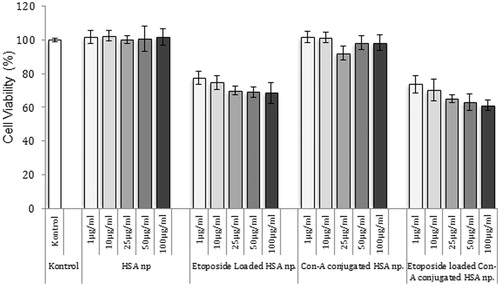
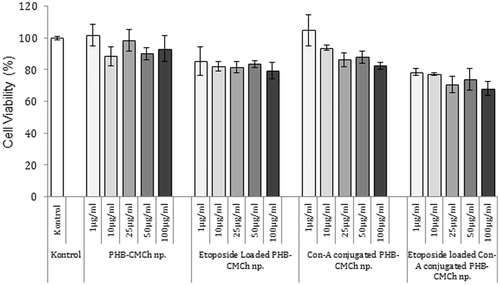
Figure 7. Cytotoxicity of different formulation of HSA nanoparticles (n = 3). The nanoparticles were incubated with MCF-7 cells for 24 h. After incubation, cell viability was measured by MTT assay. Data are expressed as percent of control mean ± SD of three independent experiments.