Figures & data
Figure 1. Schematic of Markov model. Diagram adapted from Speakman et al.Citation15.
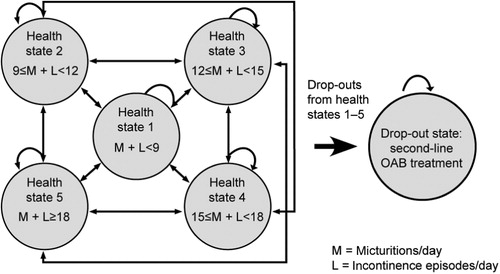
Table 1. Utility values used in the Markov model. Adapted from Kobelt et al.Citation14*.
Table 2. Unit costs used in each analysis.
Table 3. Sensitivity analyses performed.
Figure 2. Distribution of patients in health states 1–5 after treatment with solifenacin 5 mg/day or oxybutynin IR 15 mg/day, estimated over a 1-year period. See for the distribution at baseline.
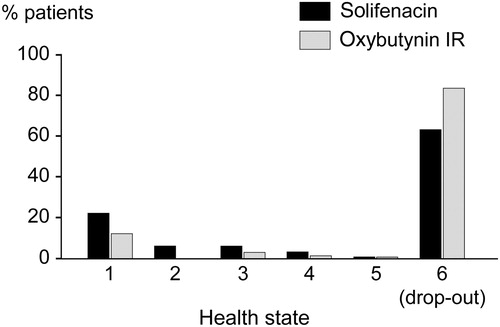
Table 4. Summary of costs, QALYs, and ICUR results at 1 year with solifenacin 5 mg/day and oxybutynin IR 15 mg/day, with and without the cost of incontinence pads. Costs are in CAN$.
Figure 3. Cost-effectiveness acceptability curve of solifenacin 5 mg/day versus oxybutynin IR 15 mg/day, with and without the cost of incontinence pads.
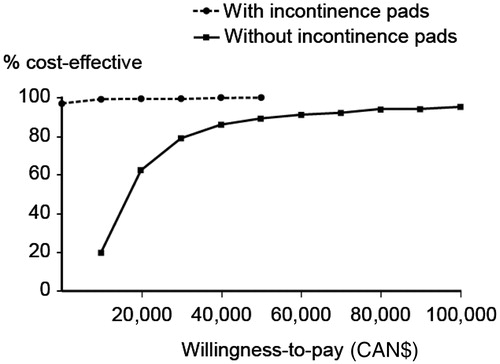
Table 5. Summary of results of sensitivity analyses, in which the cost of incontinence pads was excluded from the base case (costs in CAN$).