Figures & data
Table 1. The Joint Commission national hospital inpatient quality measures for venous thromboembolismCitation17.
Table 2. Characteristics of rivaroxaban, dabigatran, and apixaban.
Table 3. Outcomes in the EINSTEIN acute DVT and continued treatment studiesCitation33.
Figure 1. Phase III VTE treatment trial designs for apixaban: AMPLIFY (a) and AMPLIFY-EXT (b). DVT, deep vein thrombosis; INR, international normalized ratio; PE, pulmonary embolism; R, randomization; VTE, venous thromboembolism.
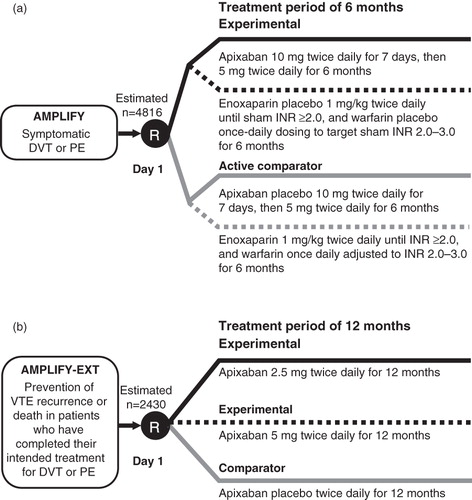
Figure 2. Phase III VTE treatment trial designs for dabigatran etexilate: RE-COVER and RE-COVER II (a), RE-MEDY (b), and RE-SONATE (c). DVT, deep vein thrombosis; INR, international normalized ratio; PE, pulmonary embolism; R, randomization; VKA, vitamin K antagonist; VTE, venous thromboembolism.
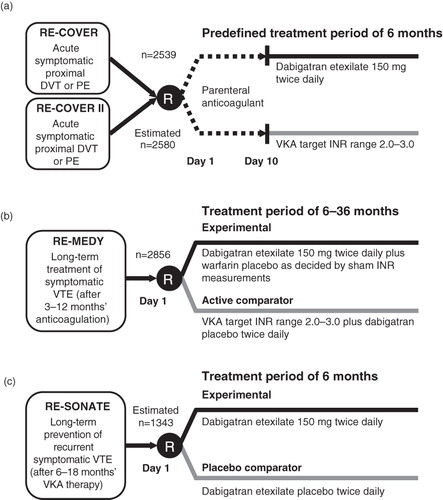
Table 4. Outcomes in the RE-COVER studyCitation40.