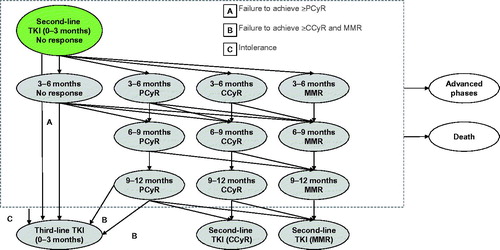
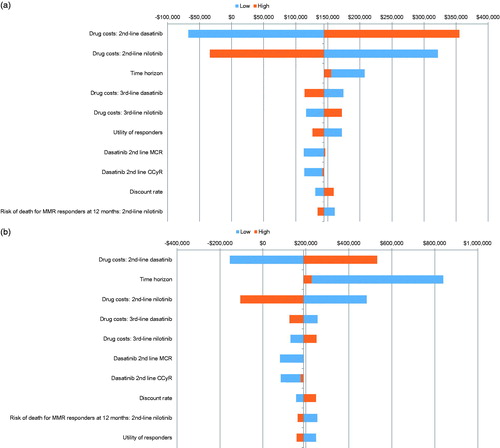
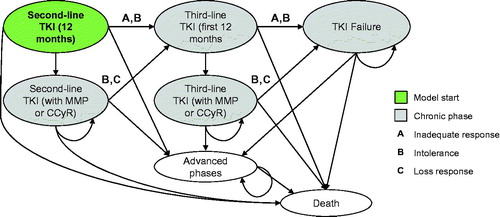
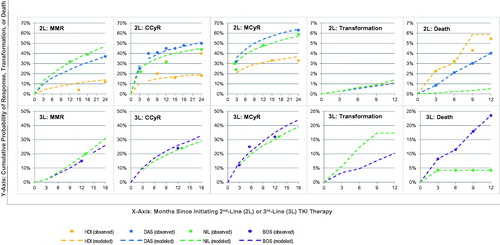
Figures & data
Table 1. TKI sequences evaluated for patient populations.
Table 3. Model results for the imatinib-resistant population.
Table 4. Model results for the imatinib-intolerant population.
Table 5. Results of scenario analyses.
Table A5. Second-line: observed and interpolated death probabilities.
Table A6. Third-line: observed and interpolated response rates on bosutinib.
Table A7. Third-line: observed and interpolated response rates on dasatinib/nilotinib.
Table A8. Third-line observed and interpolated probabilities of transformation on dasatinib/nilotinib and bosutinib.
Table A9. Third-line: observed and interpolated probabilities of death on dasatinib/nilotinib and bosutinib.
Table A1. Second-line: observed and interpolated CCyR rates.
Table A2. Second-line: observed and interpolated MCyR rates.
Table A3. Second-line: observed and interpolated MCyR rates.
Table A4. Second-line: observed and interpolated transformation probabilities.